Research Article
Molecular Modelling Analysis of the Metabolism of Aniracetam
Discipline of Biomedical Science, School of Medical Sciences, Faculty of Medicine, Cumberland Campus, C42, The University of Sydney, Lidcombe, NSW, Australia
Schizoprenia is a complex psychiatric disorder characterized by variable expression of three major categories of symptoms termed positive, negative and cognitive symptoms. Positive symptoms (also called psychotic symptoms) include hallucinations and delusions whereas negative symptoms include apathy, anhedonia and social and emotional withdrawal (Rogers and Schmidt, 2006). Schizoprenia is now viewed as being the result of a neurochemical imbalance across multiple transmitter systems (Larulle et al., 1999). It is believed that reduced glutamergic neurotransmission, particularly at the N-methyl-d-aspartate (NMDA) receptor subtype, underlies even the dopaminergic abnormalities that are common in schizophrenic patients (Javitt, 2004). This growing understanding of neurochemical deficits underlying the disease has led to a corresponding broadening in search for novel treatment approaches. Aniracetam (1-p-anisoyl-2-pyrrolidinone; ACM) is a pyrrolidinone-type cognition enhancer that has been clinically used in the treatment of behavioural and psychological symptoms of dementia following stroke and Alzheimer’s disease (Nakamuara, 2002). ACM has been found to preferentially increase extracellular levels of dopamine (DA) and serotonin in the prefrontal cortex (PFC), basolateral amugdala and dorsal hippocampus of the mesocorticolimbic system in stroke-prone spontaneously hypersensitive rats. The drug is recognized to be an allosteric modulator of α-amino-3-hydroxy-5-methyl-4-isoxazolepropanoic acid (AMPA) receptors. It is believed to block desentization of the AMPA receptors in the suprachiasmatic nucleus (SCN) that has been activated by glutamate (Lee and Benfield, 1994).
After oral administration, ACM is rapidly absorbed from the intestinal tract (Ogiso et al., 1998). The metabolites of ACM include 2-pyrrolidinone (PD), p-anisic acid (AA), 4-p-anisamidobutanoic acid (ABA), succinimide (SD), 5-hydroxy-pyrrolidinone (5HPD), succinimide (SD) (Yoshimoto et al., 2000). ACM is first metabolized to ABA, AA and PD. PD is further metabolized into 5-hydroxy-pyrrolidinone (5HPD). 5HPD is metabolized into succinimide (SD) which enters Krebs cycle producing carbon dioxide. AA is conjugated with glycine and glucuronic acid to produce AA-glycine conjugate and AA-glucuronide, respectively. It is believed that the metabolite PD enhances activity of activated Protein Kinase C (PKC), thereby potentiating α7 receptor responses and then leading to facilitation of hippocampal synaptic responses.
In this study, molecular modelling analyses have been carried out using the program Spartan ’02 (Spartan, 2002) to provide information on the relative toxicity of ACM and its metabolites.
COMPUTATIONAL METHODS
The geometries of ACM and its metabolites PD, ABA, AA and 5HPD have been optimized based on molecular mechanics (Fig. 1), semi-empirical and DFT (Density functional theory) calculations, using the molecular modelling program Spartan ’04. Molecular mechanics calculations were carried out using MMFF force field.
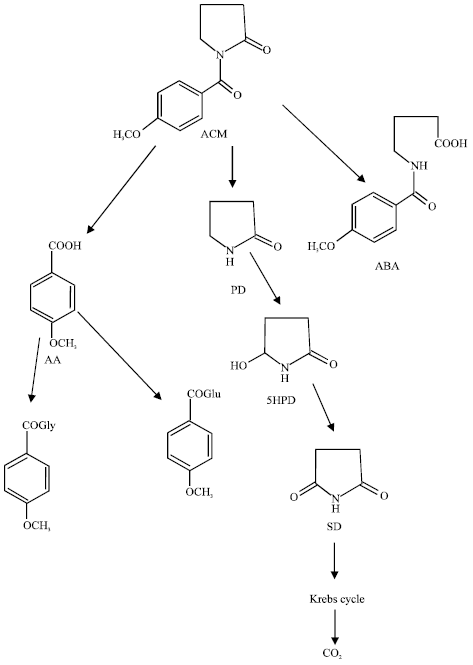 | |
| Fig. 1: | Metabolic pathways for ACM in rats and humans (Nakamura and Shirane, 1999) |
Semi-empirical calculations were carried out using the routine PM3. DFT calculations were carried at B3LYP/6-31G* level. In optimization calculations, a RMS gradient of 0.001 was set as the terminating condition. For the optimized structures, single point calculations were carried out to give heat of formation, enthalpy, entropy, free energy, dipole moment, solvation energy, energies for HOMO (Highest occupied molecular orbital) and LUMO (Lowest unoccupied molecular orbital). The order of calculations: molecular mechanics followed by semi-empirical followed by DFT ensured that the structure was not embedded in a local minimum. To further check whether the global minimum was reached, some calculations were carried out with improvable structures. It was found that when the stated order was followed, structure corresponding to the global minimum or close to that could ultimately be reached in all cases. Although RMS gradient of 0.001 may not be sufficiently low for vibrational analysis, it is believed to be sufficient for calculations associated with electronic energy levels.
Table 1 shows the total energy, heat of formation as per PM3 calculation, enthalpy, entropy, free energy, surface area, volume, dipole moment and energies of HOMO and LUMO as per both PM3 and DFT calculations for ACM and its metabolites PD, ABA, AA and 5HPD. Figure 2-6 give the regions of negative electrostatic potential (greyish-white envelopes) in (a), HOMOs (where red indicates HOMOs with high electron density) in (b), LUMOs in (c) and density of electrostatic potential on the molecular surface (where red indicates negative, blue indicates positive and green indicates neutral) in (d) as applied to optimized structures of ACM and its metabolites PD, ABA, AA and 5HPD.
| Table 1: | Calculated thermodynamic and other parameters of ACM and its metabolites |
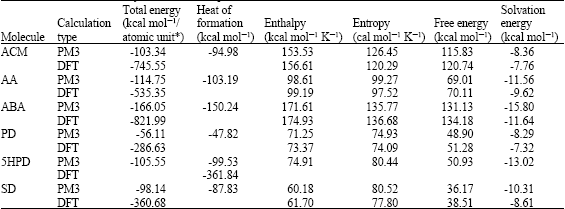 | |
| Table 1: | Continued |
-2k7-164-171.gif) | |
| *In atomic units from DFT calculations | |
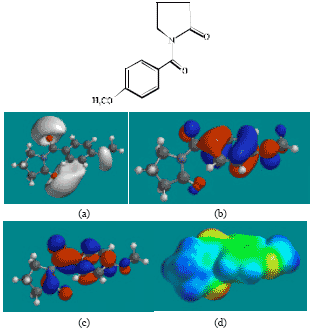 | |
| Fig. 2: | Structure of ACM giving in: (a) the electrostatic potential (greyish envelope denotes negative electrostatic potential), (b) the HOMOs, (where red indicates HOMOs with high electron density) (c) the LUMOs (where blue indicates LUMOs) and in (d) density of electrostatic potential on the molecular surface (where red indicates negative, blue indicates positive and green indicates neutral) |
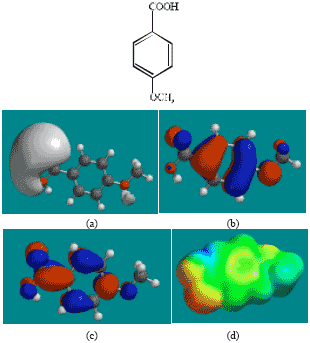 | |
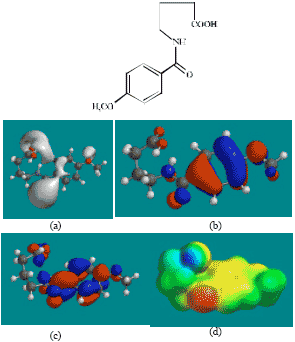
| Fig. 3: | Structure of AA giving in: (a) the electrostatic potential (greyish envelope denotes negative electrostatic potential), (b) the HOMOs, (where red indicates HOMOs with high electron density) (c) the LUMOs (where blue indicates LUMOs) and in (d) density of electrostatic potential on the molecular surface (where red indicates negative, blue indicates positive and green indicates neutral) |
 | |
| Fig. 4: | Structure of ABA giving in: (a) the electrostatic potential (greyish envelope denotes negative electrostatic potential), (b) the HOMOs, (where red indicates HOMOs with high electron density) (c) the LUMOs (where blue indicates LUMOs) and in (d) density of electrostatic potential on the molecular surface (where red indicates negative, blue indicates positive and green indicates neutral) |
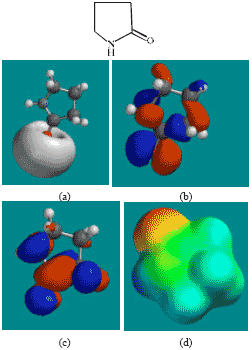 | |
| Fig. 5: | Structure of PD giving in: (a) the electrostatic potential (greyish envelope denotes negative electrostatic potential), (b) the HOMOs, (where red indicates HOMOs with high electron density) (c) the LUMOs (where blue indicates LUMOs) and in (d) density of electrostatic potential on the molecular surface (where red indicates negative, blue indicates positive and green indicates neutral) |
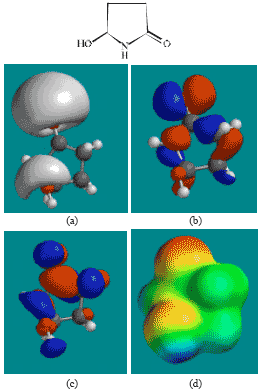 | |
| Fig. 6: | Structure of 5HPD giving in: (a) the electrostatic potential (greyish envelope denotes negative electrostatic potential), (b) the HOMOs, (where red indicates HOMOs with high electron density) (c) the LUMOs (where blue indicates LUMOs) and in (d) density of electrostatic potential on the molecular surface (where red indicates negative, blue indicates positive and green indicates neutral) |
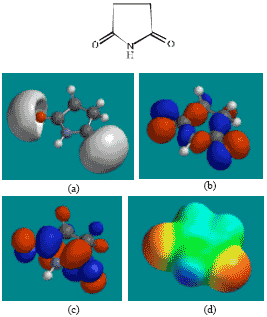 | |
| Fig. 7: | Structure of SD giving in: (a) the electrostatic potential (greyish envelope denotes negative electrostatic potential), (b) the HOMOs, (where red indicates HOMOs with high electron density) (c) the LUMOs (where blue indicates LUMOs) and in (d) density of electrostatic potential on the molecular surface (where red indicates negative, blue indicates positive and green indicates neutral) |
The LUMO-HOMO energy differences for ACM and its metabolites from DFT calculations are found to range from 4.89 to 7.4 eV, indicating that the compounds would all be kinetically inert with SD (Fig. 7) being the most inert and the parent drug ACM being the least inert one.
In the case of ACM, AA, ABA, PD, 5HPD and SD, the electrostatic potential is found to be more negative around oxygen atoms, indicating that the positions may be subject to electrophilic attack.
In the case of ACM (Fig. 2), the HOMOs with high electron density are found to be slightly more localised than the LUMOs. In the case of AA (Fig. 3), PD (Fig. 5) and 5HPD (Fig. 6), both the HOMOs with high electron density and the LUMOs are found to be close to nearly all the non-hydrogen atoms. In the case of ABA (Fig. 4), both the HOMOs with high electron density and the LUMOs are found to be close to essentially the same non-hydrogen atoms.
The overlap of HOMO with high electron density and region of negative electrostatic potential at some positions, gives further support to the idea that the positions may be subject to electrophilic attack.
The molecular surfaces of ACM and all its metabolites are found to possess significant amounts of electron-rich (yellow and red) and neutral (green) regions so that they may be subject to electrophilic and lyophilic attacks. The molecular surface of ACM is also found to possess significant amounts of electron-deficient (blue) regions so that it may be subject to nucleophilic attacks such as those due to glutathione and nucleobases in DNA. Reaction with glutathione can induce cellular toxicity by compromising the antioxidant status of the cell whereas that with nucleobases in DNA can cause DNA damage. However, as stated earlier, since ACM is expected to be moderately inert kinetically, the rate of such adverse reactions may be low unless speeded up enzymatically.
Aniracetam (ACM) is a pyrrolidinone-type cognition enhancer that has been used in the treatment of behavioural and psychological symptoms of dementia following stroke and Alzheimer’s disease. Molecular modelling analyses based on semi-empirical and DFT calculations show that both ACM and all its metabolites have large LUMO-HOMO energy differences so that they would be kinetically inert, with the parent drug being least inert. The molecular surface of the parent drug ACM is found to possess significant amounts of electron-deficient regions so that it can react with glutathione and nucleobases in DNA. However, ACM being to some extent kinetically inert, the rates of such adverse reactions may be low, unless speeded up enzymatically.
Fazlul Huq is grateful to the Discipline of Biomedical Science, School of Medical Sciences, The University of Sydney for the time release from teaching.