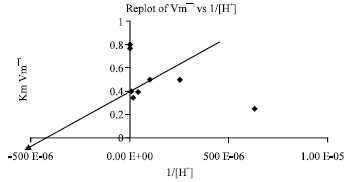
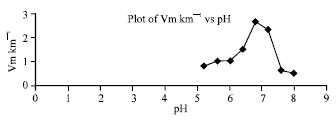
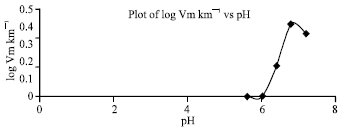
Research Article
Effect of pH on the Synthesis of L-3,4-Dihydroxyphenylalanine by Tyrosinase of Edible Mushroom
Department of Pilot Biotechnology, Pasteur Institute of Iran, Tehran 13164, Iran
A. Akbarzadeh
Department of Pilot Biotechnology, Pasteur Institute of Iran, Tehran 13164, Iran
L. Teimoori
Department of Pilot Biotechnology, Pasteur Institute of Iran, Tehran 13164, Iran
M. Nejati
Department of Bacterial Vaccine and Antigens Production, Pasteur Institute of Iran, Tehran 13164, Iran
A. Faranghi
Department of Pilot Biotechnology, Pasteur Institute of Iran, Tehran 13164, Iran