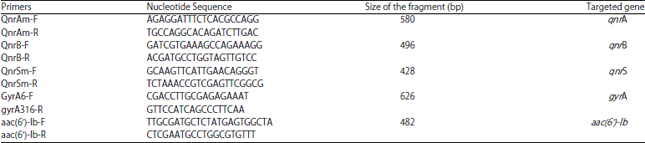
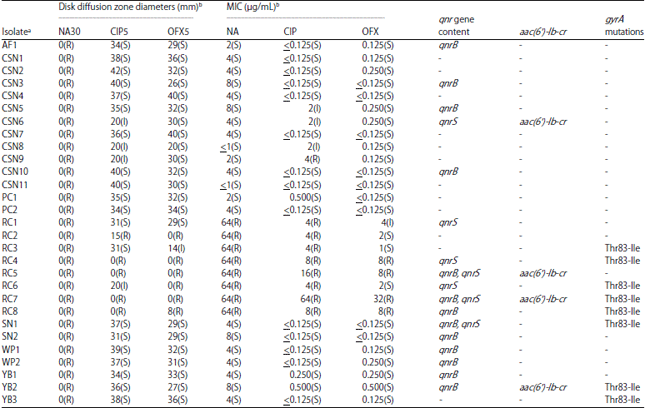
Research Article
High Prevalence of Quinolone Resistance Genes in Citrobacter freundii Isolated from Pet Turtles
Veterinary Medical Center, College of Veterinary Medicine, Chungbuk National University, Chungdae-ro 1, Seowon-gu, 28644 Cheongju, Chungbuk, Korea
B.C.J. De Silva
Veterinary Medical Center, College of Veterinary Medicine, Chungbuk National University, Chungdae-ro 1, Seowon-gu, 28644 Cheongju, Chungbuk, Korea
S.H.M.P. Wimalasena
Veterinary Medical Center, College of Veterinary Medicine, Chungbuk National University, Chungdae-ro 1, Seowon-gu, 28644 Cheongju, Chungbuk, Korea
H.N.K.S. Pathirana
Veterinary Medical Center, College of Veterinary Medicine, Chungbuk National University, Chungdae-ro 1, Seowon-gu, 28644 Cheongju, Chungbuk, Korea
Gang-Joon Heo
Veterinary Medical Center, College of Veterinary Medicine, Chungbuk National University, Chungdae-ro 1, Seowon-gu, 28644 Cheongju, Chungbuk, Korea
LiveDNA: 82.16116