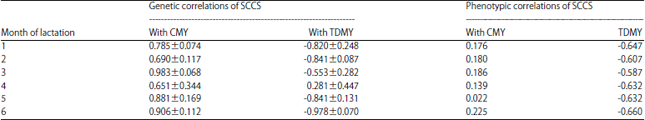
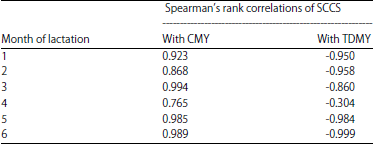
Research Article
Test Day Milk Yield as a Selection Criterion to Improve Milk Yield Based on Measures of Somatic Cells in Holstein Cow’s Milk
Department of Animal Husbandry and Wealth Development, Faculty of Veterinary Medicine, Mansoura University, El-Gomhouria Street, 35516 Mansoura, Dakahlia, Egypt
Eman A. ABO Elfadl
Department of Animal Husbandry and Wealth Development, Faculty of Veterinary Medicine, Mansoura University, El-Gomhouria Street, 35516 Mansoura, Dakahlia, Egypt