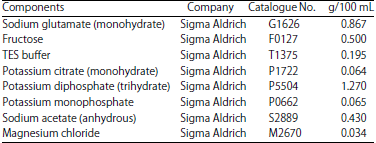
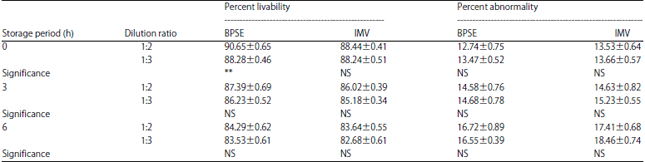
Research Article
Effect of Semen Diluents, Dilution Rates and Storage Periods on Live and Abnormal Spermatozoa of Pearl Guinea Fowls
Department of Poultry Science, Madras Veterinary College, Chennai-600 007, India
A.V. Omprakash
Department of Poultry Science, Madras Veterinary College, Chennai-600 007, India
K. Premavalli
Department of Poultry Science, Madras Veterinary College, Chennai-600 007, India