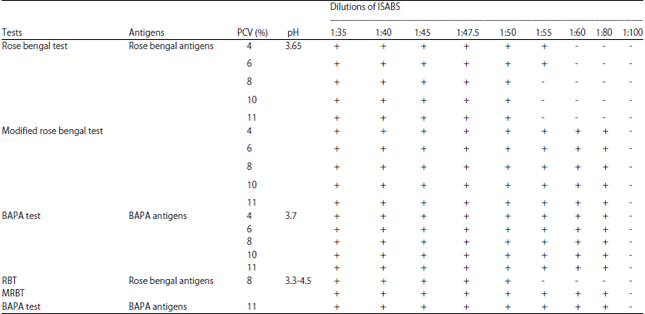
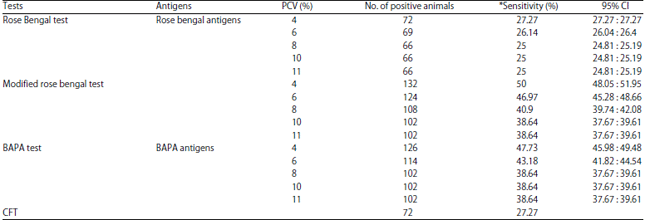
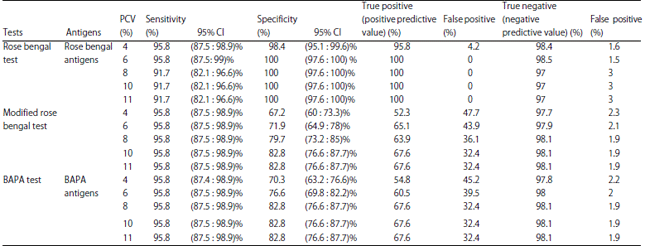
Research Article
Efficiency of Different Preparations of Rapid Slide Agglutination Antigens for the Diagnosis of Bovine and Ovine Brucellosis
Veterinary Serum and Vaccine Research Institute (VSVRI), Cairo, Egypt
Wafaa R. Abdel Aziz
Veterinary Serum and Vaccine Research Institute (VSVRI), Cairo, Egypt
Waleed S. Shell
Central Laboratory for Evaluation of Veterinary Biologics (CLEVB), Cairo, Egypt
A.A. Samy
Department of Microbiology and Immunology, National Research Center (NRC), Cairo, Egypt