Research Article
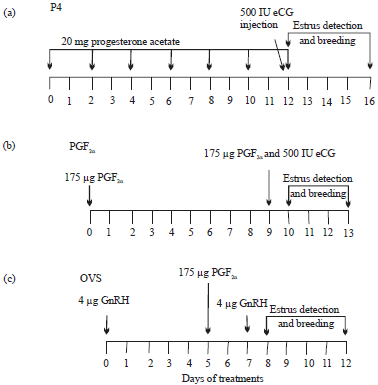
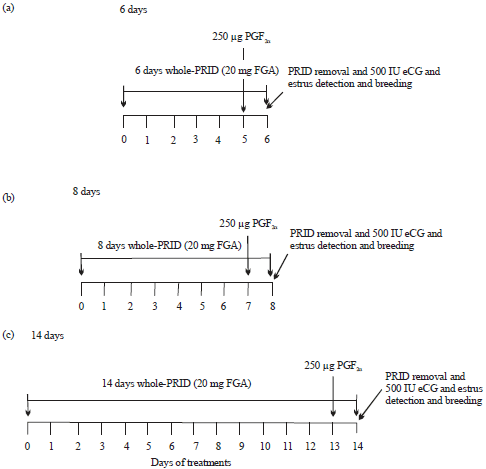
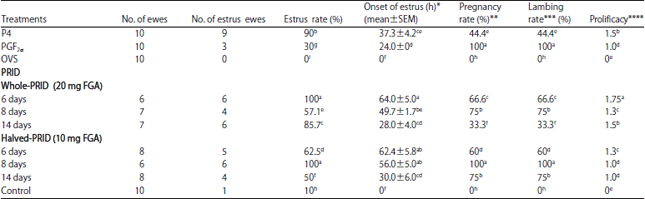
Efficacy of Various Synchronization Protocols on the Estrus Behavior, Lambing Rate and Prolificacy in Rahmani Egyptian Ewes During the Non-Breeding Season
Department of Theriogenology, Faculty of Veterinary Medicine, Kafrelsheikh University, El-Geish Street, Kafrelsheikh, 33516, Egypt
LiveDNA: 20.11979
Mohey Ashour
Riwina Animal Production Station, Agriculture production sector, Agricultural Research Center, Kafrelsheikh, 33516, Egypt
Ismail El-Kon
Department of Theriogenology, Faculty of Veterinary Medicine, Kafrelsheikh University, El-Geish Street, Kafrelsheikh, 33516, Egypt
Bassiouni Heleil
Department of Theriogenology, Faculty of Veterinary Medicine, Kafrelsheikh University, El-Geish Street, Kafrelsheikh, 33516, Egypt
El-Sayed Fattouh
Department of Theriogenology, Faculty of Veterinary Medicine, Kafrelsheikh University, El-Geish Street, Kafrelsheikh, 33516, Egypt