Review Article
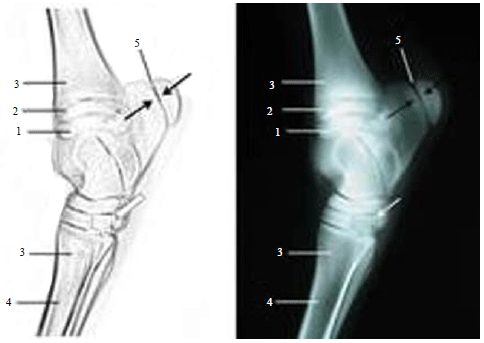
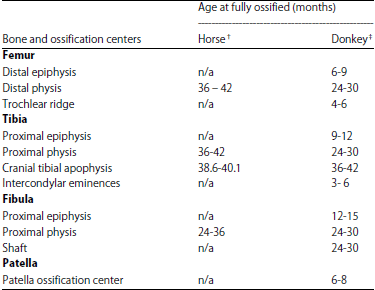
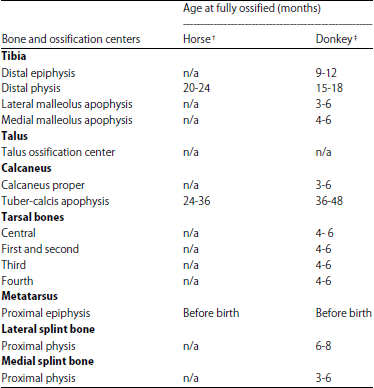
Radiographic Specification of Skeletal Maturation in Donkeys: Defining the Ossification Time of Donkey Growth Plates for Preventing Irreparable Damage
Department of Surgery, Anesthesiology and Radiology, Faculty of Veterinary Medicine, Zagazig University, El-Sharkia, Egypt
LiveDNA: 20.11794
Mudasir Bashir Gugjoo
Division of Surgery, Indian Veterinary Research Institute, Izatnagar, India
Alaa Ghazy
Department of Surgery, Anesthesiology and Radiology, Faculty of Veterinary Medicine, Kafrelsheikh University, Kafrelsheikh, Egypt
LiveDNA: 20.1024
Mohamed Gomaa
Department of Surgery, Anesthesiology and Radiology, Faculty of Veterinary Medicine, Zagazig University, El-Sharkia, Egypt
Ahmed Abdelaal
Department of Animal Medicine, Faculty of Veterinary Medicine, Zagazig University, El-Sharkia, Egypt
Amarpal
Division of Surgery, Indian Veterinary Research Institute, Izatnagar, India
LiveDNA: 91.5019
Ahmed Behery
Department of Surgery, Anesthesiology and Radiology, Faculty of Veterinary Medicine, Zagazig University, Ee-Sharkia, Egypt
Abdel-Basit Abdel-Aal
Department of Surgery, Anesthesiology and Radiology, Faculty of Veterinary Medicine, Zagazig University, El-Sharkia, Egypt
Mohamed-Tayiser Samy
Department of Surgery, Anesthesiology and Radiology, Faculty of Veterinary Medicine, Zagazig University, El-Sharkia, Egypt
Kuldeep Dhama
Division of Pathology, ICAR-Indian Veterinary Research Institute, Izatnagar, India
LiveDNA: 91.4710