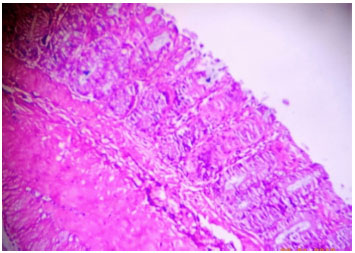
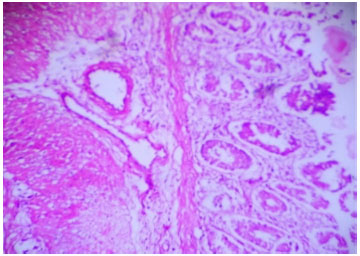
Research Article
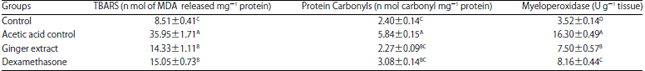
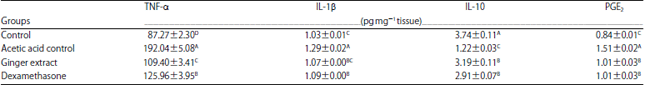
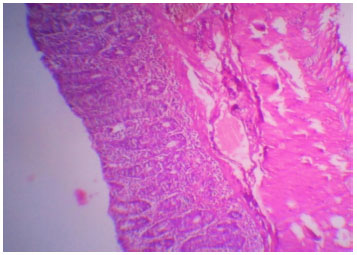
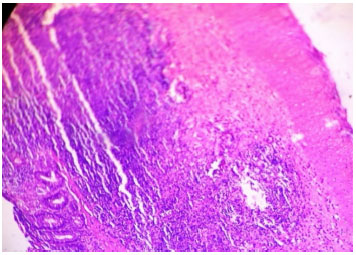
Ameliorative Effect of Ginger Extract in Acetic Acid Induced Ulcerative Colitis in Rats
Department of Veterinary Pharmacology and Toxicology,
Matukumalli Usharani
Department of Veterinary Pharmacology and Toxicology,
Boobalan Gopu
Department of Veterinary Pharmacology and Toxicology,
LiveDNA: 91.14669
Alla Gopala Reddy
Department of Veterinary Pharmacology and Toxicology,
Matham Vijay Kumar
Department of Veterinary Pharmacology and Toxicology,
Chitturi Sree Venkat Satish Kumar
Department of Veterinary Pharmacology and Toxicology,
Doppalapudi Madhuri
Department of Veterinary Pathology, College of Veterinary Science, Rajendranagar, Hyderabad, 500030, India