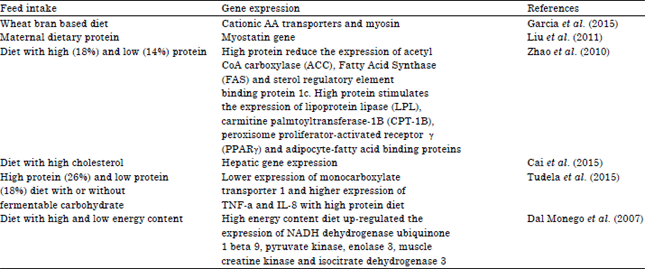
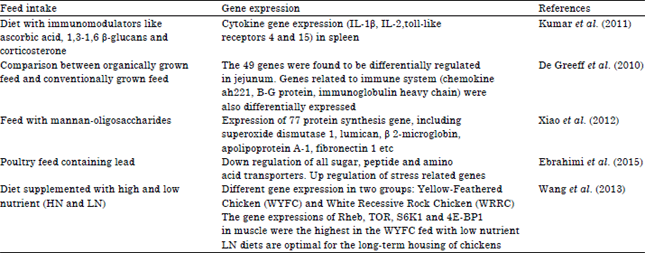
Review Article
Applications of Nutrigenomics in Animal Sectors: A Review
Department of Zoology, Visva-Bharati University, Santiniketan, 73123,5 India
Rounak Pal
Department of Zoology, Visva-Bharati University, Santiniketan, 73123,5 India
Arun Kumar Ray
Department of Zoology, Visva-Bharati University, Santiniketan, 73123,5 India