Research Article
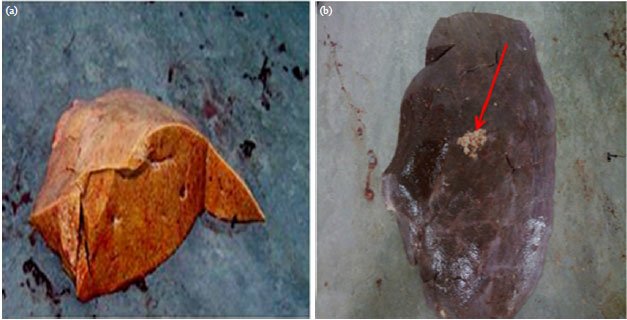
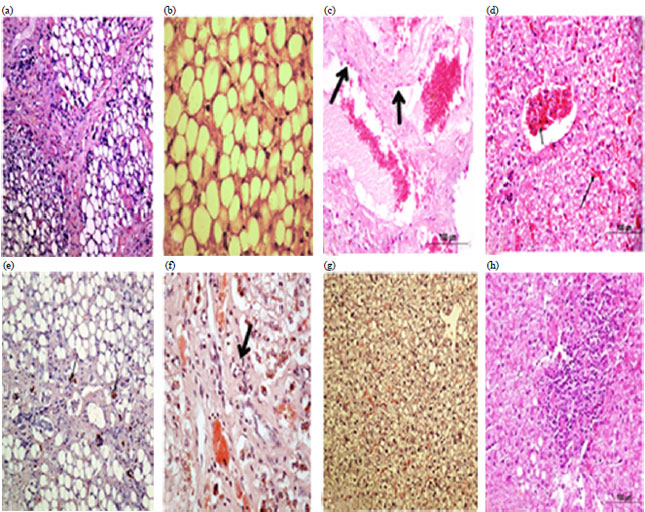
Effect of Aflatoxin B1 (AFB1) Residues on the Pathology of Camel Liver
College of Veterinary Medicine and Animal Resources, King Faisal University, Kingdom of Saudi Arabia
N. A.M. Al-Gabri
College of Veterinary Medicine and Animal Resources, King Faisal University, Kingdom of Saudi Arabia
S. E.M. Barakat
College of Veterinary Medicine and Animal Resources, King Faisal University, Kingdom of Saudi Arabia