Research Article
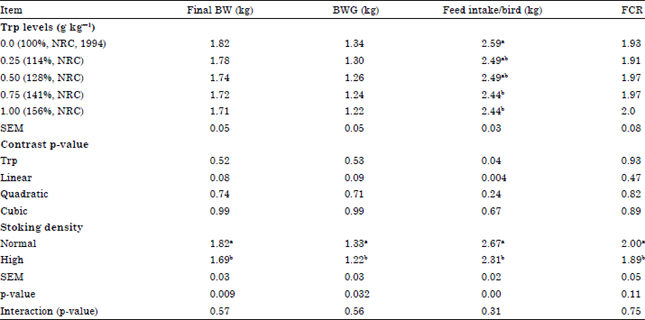
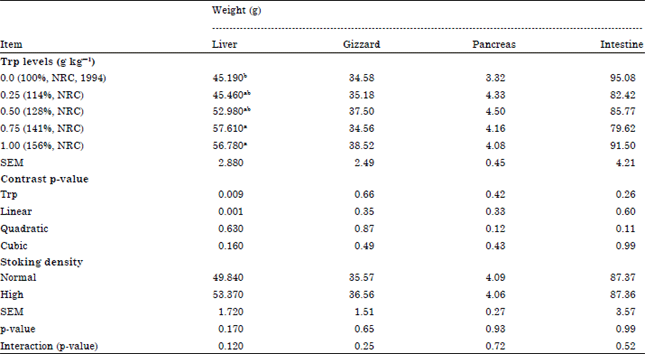
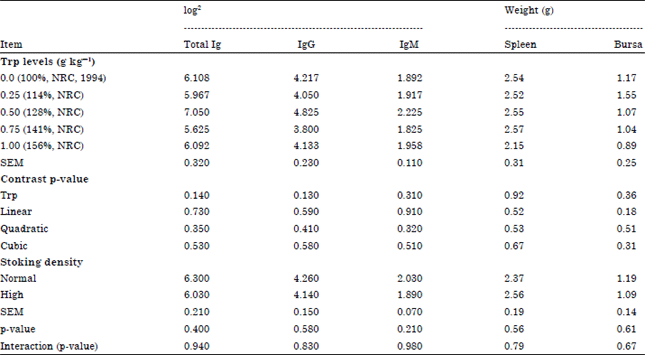
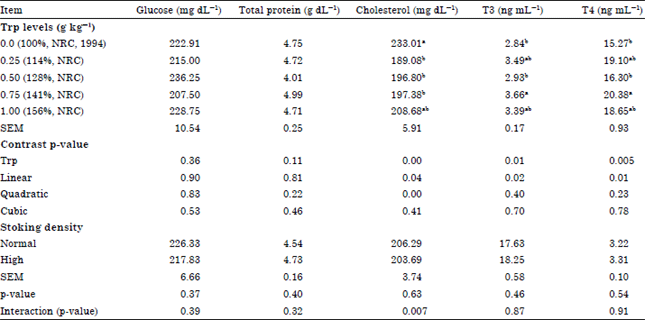
Effects of Dietary Tryptophan Levels and Stocking Density During the Growing-Finishing Phase on Broiler Performance and Immunity
Department of Poultry Production, Faculty of Agriculture, Mansoura University, Mansoura, 35516, Egypt
M.M. Azzam
Department of Poultry Production, Faculty of Agriculture, Mansoura University, Mansoura, 35516, Egypt