ABSTRACT
This study was conducted to determine the effects of dietary microencapsulated n-3 fatty acid supplementation at different concentrations on growth performance, nutrient digestibility, blood profiles, carcass characteristics and fatty acids compositions of muscle and adipose tissue in finishing pigs. Insufficient and unbalanced amount of essential fatty acids may cause a number of diseases such as atherosclerosis, cardiovascular diseases but high consumption improves growth performance, brain development and disease resistance. A total of 150 pigs with initial Body Weights (BW) of 55.66±1.42 kg were used in the 12 week experiment. Pigs were randomly allotted to 3 dietary treatments with 10 pens per treatment and 5 pigs per pen. The three dietary treatments included: (1) CON (basal diet), (2) FA15 (CON+1.5% n-3 fatty acid) and (3) FA30 (CON+3.0% n-3 fatty acid). Results of the whole experimental period showed that additions of 1.5 and 3.0% of n-3 fatty acid to the diet did not affect (p>0.05) growth performance, nutrient digestibility and blood characteristics in finishing pigs. The Average Daily Gain (ADG) was 724, 743 and 754 g in CON, FA15 and FA30, respectively at the end of experiment. Pigs fed the FA30 treatment diet had a decreased (p<0.05) Longissimus Muscle (LM) area compared with those fed the FA15 treatment diets. Palmitic acid percentage of LM was decreased (p<0.05) by the FA15 and FA30 dietary treatments compared with the CON dietary treatment. Total n-3/n-6 ratio of both LM and subcutaneous adipose (s.c. adipose), docosahexaenoic acid (DHA) and total n-3 of s.c. Adipose in the FA15 and FA30 dietary treatments were greater (p<0.05) than those in the CON dietary treatment. In conclusion, there were no observed effects on growth performance, nutrient digestibility and carcass characteristics in finishing pigs associated with microencapsulated n-3 fatty acid supplementation.
PDF Abstract XML References Citation
How to cite this article
DOI: 10.3923/ajava.2013.703.712
URL: https://scialert.net/abstract/?doi=ajava.2013.703.712
INTRODUCTION
Essential fatty acids are molecules that cannot be synthesized by humans and animals body but are vital for normal metabolism (Pollan, 2007). Essential fatty acids play a part in many metabolic processes and there is evidence to suggest that low levels of essential fatty acids, or an essential fatty acid imbalance, may be a factor into a number of illnesses (Kruger and Horrobin, 1997). n-3 and n-6 fatty acids are the two families of essential fatty acids. The n-3 fatty acids which are important in human nutrition are: α-linolenic acid (18:3 n-3, ALA), eicosapentaenoic acid (20:5 n-3, EPA) and docosahexaenoic acid (22:6 n-3, DHA). The human body can convert ALA to EPA and subsequently DHA, all of which are polyunsaturated fatty acids (PUFA). According to Dyerberg et al. (1975), atherosclerosis, blood pressure, heart rates and triglycerides were low in the member of Inuit tribe due to consumption of high level of n-3 fatty acids. Furthermore, n-3 fatty acids have beneficial health effects on humans, especially in the control of cardiovascular diseases (Dewailly et al., 2001), support of normal brain growth and development (Edwards et al., 1998) and decreased inflammation of joint pain (Goldberg and Katz, 2007). High intakes of EPA and DHA may improve disease resistance in pigs (Irie and Sakimoto, 1992; Calder, 1996; Jolly et al., 1997), which may have beneficial growth performance effects (Abril et al., 2003). It is suggested that growth performance could be restricted by insufficient PUFA intake. Otherwise, Jaturasitha et al. (2009) reported that n-3 fatty acids from tuna oil had no significant effect on the growth performance of fattening pigs. Coates and Ayerza (2009) reported that growth performance was not affected by dietary treatments in finishing pigs supplemented at levels of 10 and 20% of Chia seed (Salvia hispanical). No effects on growth performance were observed in growing pigs by ALA supplementation from whole crushed linseed (Kouba et al., 2003). Taken together, the effects of n-3 fatty acids on growth performance in pigs are not always consistent. Due to the important roles that essential fatty acids play in our life, additional research needs to be conducted. Some studies reported the method and effect of microencapsulation (Piva et al., 1997; Kierczynska, 1988; Walz and Pallauf, 1988). Piva et al. (2007) stated that lipid microencapsulation blend reducing the faster disappearance of active ingredients (organic acids and natural identical flavors) exciting in the stomach. The objective of this experiment was to evaluate the effects of dietary microencapsulated n-3 supplementation on growth performance, nutrient digestibility, blood profiles, carcass characteristics and fatty acid compositions in finishing pigs.
MATERIALS AND METHODS
The experimental protocols used were approved by the Animal Care and Use Committee of Dankook University.
Experimental design, animals and housing: A total of 150 crossbred (LandracexYorkshirexDuroc; 95 day age) pigs with an average initial Body Weight (BW) of 55.66±1.42 kg were used in this 12 week experiment. At the beginning of the experiment, pigs were allotted to one of 3 dietary treatments on the basis of initial BW. There were 5 pigs per pen and 10 pens per treatment. The dietary treatments were: (1) CON (basal diet), (2) FA15 (CON+1.5% n-3 fatty acid) and (3) FA30 (CON+3.0% n-3 fatty acid). Microencapsulated n-3 fatty acid contains 92% α-linolenic acid from linseed oil, 5% DHA and 3% EPA from fish oil. Diets (Table 1) were formulated to meet or exceed the requirements recommended by the NRC (1998). Pigs were housed in an environmentally-controlled; slatted-floor facility in 30 adjacent pens (1.8x1.8 m), room temperature was maintained at approximately 24°C. Each pen was equipped with a self-feeder and nipple waterer to allow ad libitum access to feed and water throughout the experiment.
Sampling and measurements: Pig BW and feed consumption were measured at the end of week 6 and 12 to monitor Average Daily Gain (ADG), Average Daily Feed Intake (ADFI) and gain:feed ratio (G:F).
| Table 1: | Composition of the experimental diets (as-fed basa) |
 | |
| aSupplied per kg diet: 4,000 IU vitamin A, 800 IU vitamin D3, 171 IU vitamin E, 2 mg vitamin K, 4 mg vitamin B2, 1 mg vitamin B6, 16 mg vitamin B12, 11 mg pantothenic acid, 20 mg niacin and 0.08 mg biotin, bSupplied per kg diet: 220 mg Cu, 175 mg Fe, 191 mg Zn, 89 mg Mn,0.3 mg I, 0.5 mg Co and 0.4 mg Se | |
Chromium oxide was added to the diets as an indigestible marker at 0.20% of the diet for 7 days before fecal collection at 6 and 12 weeks for calculation of Dry Matter (DM), Nitrogen (N) and energy digestibility. Fecal grab samples were collected at random from at least two pigs in each pen. All feed and fecal samples were stored at -20°C until analysis. Before chemical analysis, fecal samples were thawed at 54°C for 72 h freeze-dried and then finely ground to pass through a 1 mm screen. All feed and fecal samples were analyzed for DM and N (AOAC, 1995). Chromium levels were determined via UV absorption spectrophotometry (UV-1201, Shimadzu, Kyoto, Japan).
At week 6 of the experiment, blood samples were collected at the cervical vein into both K3EDTA vacuum tubes and nonheparinized vacuum tubes (Becton Dickinson Vacutainer Systems, Franklin Lakes, NJ, USA) from two pigs in each pen; the same pigs were then sampled at week 12 of the experiment. After collection, the serum samples from the nonheparinized vacuum tubes were centrifuged at 2,000xg for 30 min at 4°C. The concentrations of total cholesterol, High-density Lipoprotein (HDL) cholesterol, Low-density Lipoprotein (LDL) cholesterol and triglycerides in the serum samples were analyzed with an automatic biochemical analyzer (RA-1000, Bayer Corp., Tarrytown, NY) using colorimetric methods. Red Blood Cell (RBC), White Blood Cell (WBC) and lymphocyte counts in the whole blood were analyzed using an automatic blood analyzer (ADVIA 120, Bayer, NY).
At the end of the experiment, all of the pigs were transferred to a slaughterhouse. Carcasses were chilled at 2°C for 24 h and a piece of the right loin was obtained through a perpendicular cut between the 10th and 11th ribs. Before the meat quality evaluation was performed, meat samples were thawed at room temperature. Sensory evaluation (color, marbling and firmness scores) was evaluated according to National Pork Producers Council (NPPC, 1991) standards. At the same time, duplicate pH values of each sample were measured by pH meter (Fisher Scientific, Pittsburgh, PA, USA). The color measurement of lightness (L*), redness (a*) and yellowness (b*) values were determined by Minolta CR410 Chromameter (Konica Minolta Sensing, Inc., Osaka, Japan). The Water Holding Capacity (WHC) of Longissimus Muscle (LM) was measured according to the methods of Kauffman et al. (1986). A 0.2 g sample was pressed at 3,000xg for 3 min on a 125 mm-diameter piece of filter paper. The areas of the pressed sample and expressed moisture were delineated and then determined with a digitizing area-line sensor (MT-10S; M.T. Precision Co. Ltd., Tokyo, Japan). The ratio of water:meat areas was calculated, giving a measure of WHC (the smaller ratio indicates a higher WHC). Drip loss was measured using approximately 2 g of meat sample according to the plastic bag method, which was described by Honikel (1998). The sample weight was measured at d 1, 3, 5 and 7 L. Marea was measured by tracing the LM surface at the 10th rib and this was accomplished using the above-mentioned digitizing area-line sensor.
To determine the fatty acid composition of LM and subcutaneous adipose (s.c. adipose), tissue samples collected were extracted using a chloroform: methanol (3:1, v/v) mixture according to the method described by Folch et al. (1957). Next, 20 to 25 mg of the extracted fat was saponified with 0.5 M methanolic sodium hydroxide and then methylated with borontrifluoride in methanol using the method described by Metcalfe et al. (1966). The fatty acid methyl esters obtained were then separated and analyzed by gas chromatography. The fatty acid content was determined using a gas chromatographer, the HP6890 (Agilent, Waldbronn, Germany) equipped with a flame ionization detector and an HP 19091 to 136 capillary column (60 mx0.25 mm internal diameter) with a film thickness (0.25 μm) of the stationary phase. Helium was used as a carrier gasat a follow rate of 1.2 mL min-1. Oven temperature was programmed as follows: from 140 to 160°C at 1.50°C min-1, from 160 to 180°C at 0.50°C min-1 and from 180 to 230°C at 2.50°C min-1. The other chromatographic conditions were as follows: injector and detector temperatures, 280°C; sample volume injected, 1 μL. Fatty acids were identified by matching their retention times with those of their relative standards, as well as through mass spectrometry (HP5973, Agilent) of each peak.
Statistical analysis: All data were subjected to the GLM procedures of SAS (1996) as a randomized complete block design, with the pen being defined as the experimental unit. Differences among all treatments were separated by Duncan’s multiple range test. A probability level of p<0.05 was considered to be statistically significant.
RESULTS
Growth performance and apparent total tract nutrient digestibility: The effects of dietary n-3 supplementation on growth performance in finishing pigs are shown in Table 2. No effects (p>0.05) of dietary treatments on ADG, ADFI and G:F were observed during 0 to 6 week, 7 to 12 week and the overall experiment period. The highest G:F value (p>0.05) was observed in the FA 30 treatment (0.397) that was followed by 0.374 and 0.368 in the FA15 and CON treatments, respectively at 84 days. There were also no differences on Apparent Total Tract Digestibility (ATTD) of DM, N and energy among the dietary treatments (Table 3).
Blood profiles: The total cholesterol, HDL cholesterol, LDL cholesterol, triglyceride, RBC, WBC and lymphocyte counts were not affected during 0 to 6 week and 7 to 12 week by any dietary treatments (Table 4). The total cholesterol (mg dL-1) in blood was 92.12, 90.35 and 87.00 in FA30, CON and FA15, respectively (p>0.05). Table 4 showed that CON fed pigs had higher (p>0.05) HDL cholesterol in blood (42.00 mg dL-1) than FA30 and FA15 (40.75 and 37.75 mg dL-1), numerically.
| Table 2: | Effects of dietary n-3 supplementation on growth performance in finishing pigsa |
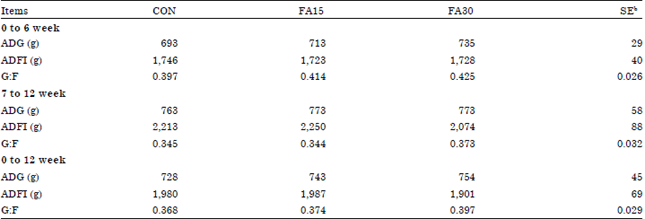 | |
| aCON, basal diet, FA15, basal diet+1.5% n-3 fatty acid, FA30, basal diet+3.0% n-3 fatty acid, bStandard error | |
| Table 3: | Effects of dietary n-3 supplementation on nutrient digestibility in finishing pigsa |
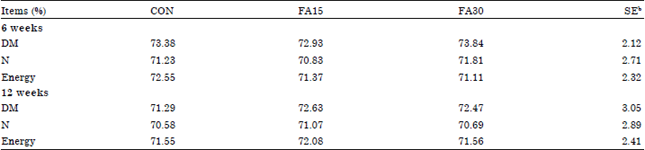 | |
| aCON, basal diet, FA15, basal diet+1.5% n-3 fatty acid, FA30, basal diet+3.0% n-3 fatty acid, bStandard error | |
| Table 4: | Effects of dietary n-3 supplementation on blood profiles in finishing pigsa |
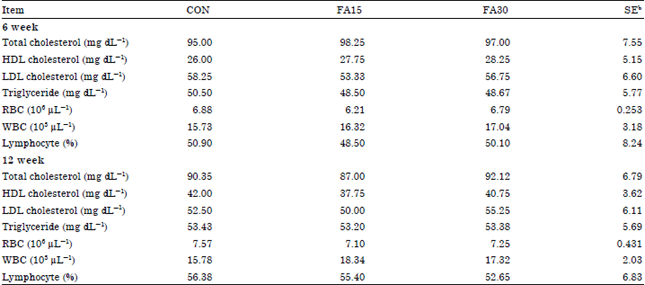 | |
| aCON, basal diet, FA15, basal diet+1.5% n-3 fatty acid, FA30, basal diet+3.0% n-3 fatty acid, bStandard error | |
Carcass characteristics: No differences were observed on sensory evaluation, meat color, pH, WHC and drip loss by the dietary treatments (Table 5). Pigs fed the FA30 treatment diets had decreased (p<0.05) LM area compared with those fed the FA15 treatment diets.
| Table 5: | Effects of dietary n-3 supplementation on carcass characteristics in finishing pigsa |
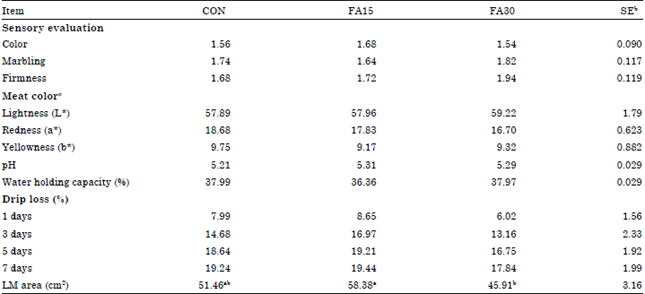 | |
| a,bMeans in the same row with difference superscripts differ (p<0.05), aCON, basal diet, FA15, basal diet+1.5% n-3 fatty acid, FA30, basal diet+3.0% n-3 fatty acid, bStandard error, cLightness: Measure of lightness to darkness (larger number indicates a lighter color), Redness: Measure of redness (larger number indicates a more intense red color) and Yellowness: Measure of yellowness (larger number indicates more yellow color) | |
| Table 6: | Effects of dietary n-3 supplementation on fatty acid composition of longissimus muscle in finishing pigs |
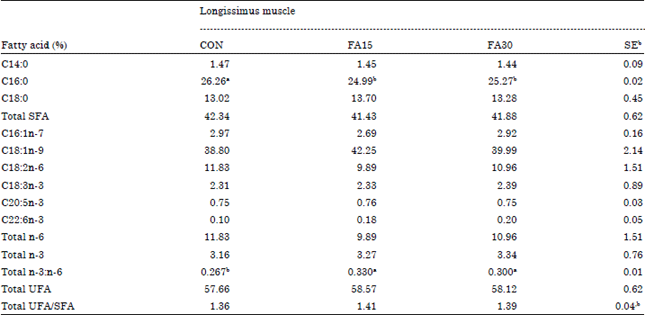 | |
| Means in the same row with difference superscripts differ (p<0.05), aCON, basal diet, FA15, basal diet+1.5% n-3 fatty acid, FA30, basal diet+3.0% n-3 fatty acid, bStandard error | |
Fatty acid composition of LM and s.c. adipose: Palmitic acid percentage of LM was decreased (p<0.05) by the FA15 and FA30 dietary treatments compared with the CON dietary treatment (Table 6). DHA and total n-3 percentage of s.c. adipose were improved (p<0.05) by the FA15 and FA30 dietary treatment diets (Table 7). Otherwise, the total ratio of n-3: n-6 of LM and s.c. adipose in the FA15 and FA30 dietary treatments was greater (p<0.05) than that in the CON dietary treatment.
| Table 7: | Effects of dietary n-3 supplementation on fatty acid composition of subcutaneous adipose in finishing pigsa |
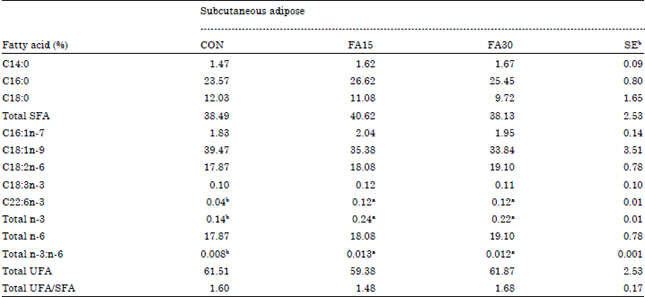 | |
| a,bMeans in the same row with difference superscripts differ (p<0.05), aCON, basal diet, FA15, basal diet+1.5% n-3 fatty acid, FA30, basal diet+3.0% n-3 fatty acid, bStandard error | |
There were no differences on other fatty acid compositions of LM and s.c. adipose among the dietary treatments.
DISCUSSION
Previous studies reported that ADG (linearly) and G:F ratios (quadratically) were increased with increasing levels of Ascophyllum nodosum seaweed extract (ANODE) in young pigs at 24 day of age (Turner et al., 2002). Abril et al. (2003) reported that pigs receiving the highest level of marine algae (5.75 kg per pig over a 42 day trial period) showed improved ADG. However, He et al. (2002) reported that ADFI, ADG and Feed Conversion Ratio (FCR) were not significantly different when Laminaria digitata algae were supplemented. Gardiner et al. (2008) observed that dietary supplementation of 3, 6 g and 9 g ANE kg-1 supplementation linearly decreased ADG while having and no effect on FI or/and FCR in grower-finisher pigs. In addition, Sardi et al. (2006) reported that dietary supplementation with DHA-rich marine algae (Schizochytrium sp.) on Italian heavy pigs did not produce significantly different ADG or/and FCR. In our study, no significant differences were observed in growth performance at any period. Those results supported the body of existing literature and research within the area mentioned above He et al. (2002), Sardi et al. (2006), Gardiner et al. (2008), However, there were contrasting reports on growth performance. Those differences may be caused by different experimental variables such as the resources of n-3 fatty acids, dosage of n-3 fatty acids, pig growth phases and environment conditions.
Gardiner et al. (2008) reported that a dietary supplementation of 2.5 g ANE kg-1 had no effect on the ATTD of DM, N and energy in grower-finisher pigs which was consistent with our results on the ATTD of DM, N and energy with a dietary supplementation of n-3 fatty acid in finishing pigs. However, Hintz et al. (1966) reported that when pelleted rations containing no Chlorella spp., 6 or 10% Chlorella spp. were fed to barrows (40 to 46 kg), DM digestibility was higher in pigs not fed a Chlorella spp. diet than pigs fed a diet containing 6% or 10% Chlorella spp. We suggest that n-3 fatty acid supplementation may have no effect on digestibility in finishing pigs.
Jaturasitha et al. (2009) reported that WHC and marbling of the loin as well as and firmness remained unaffected and a lighter, less red and less yellow backfat were observed in fattening pigs by n-3 fatty acid supplementation derived from 1 or 3% of tuna oil in lean and lipid tissues. However, Sardi et al. (2006) reported that there were no significant differences on carcass characteristics in heavy pigs whose diet was supplemented with 2.5 and 5% marine algae (Schizochytrium). Corino et al. (2002) also reported that the loin composition of heavy pigs fed diets containing either tallow or/and corn oil and rapeseed oil (all 2.5%) during the finishing phase did not vary significant. In our study, there were no observed differences in marbling, firmness, lightness, redness, yellowness, pH, WHC, or/and drip loss which were agreed with the results of Corino et al. (2002) and Sardi et al. (2006). However, our research indicated that LM area was decreased with the increasing levels of n-3 fatty acid supplementation in our research.
Coates and Ayerza (2009) reported that significantly less palmitic acid was found in the s.c. adipose tissue with Chia seed treatments at concentrations of 10 and 20% in finishing pigs and this was supported by our study findings regarding palmitic acid. Additionally, the total n-3: n-6 ratio of s.c. adipose increased in FA5 and FA30 treatments compared with CON. Pigs with an initial weight of approximately 78 kg who were fed an extruded linseed supplement diet displayed a decreased n-6:n-3 PUFA ratio, although linseed was used as a n-3 fatty acid source (Corino et al., 2008). The n-3:n-6 ratio has been relevant to the competition for synthesis of longer-chain PUFA between each series. Irie and Sakimoto (1992) demonstrated that EPA and DHA of fat can be elevated greatly by feeding fish oil to pigs for 2 to 4 week before slaughter. Jaturasitha et al. (2002) reported that 1% tuna oil in the diet was observed to have the effect of enriching total n-3 fatty acid, EPA and DHA of lean and lipid tissue throughout fattening period. The increased total n-3 fatty acid and DHA of s.c. adipose were also found in our study. However, Bryhni et al. (2002) demonstrated that the use of 0.2 to 0.4% fish oil had only minor effects. Taken together, the present study results indicate that the n-3 fatty acid composition of s.c. adipose can be increased by supplementing with n-3 fatty acid.
CONCLUSION
Results indicate that microencapsulated n-3 fatty acid supplementation potentially has no effect on growth performance, nutrient digestibility or carcass characteristics in finishing pigs. Pigs fed microencapsulated n-3 fatty acid supplementation diet had an increased n-3 fatty acid composition of s.c. adipose. However, more research is needed to study the effects of microencapsulated n-3 fatty acid supplementation on growth performance and determine the optimal amount of n-3 fatty acid to improve the fatty acid in finishing pigs.
ACKNOWLEDGMENTS
This work was carried out with the support of "Cooperative Research Program for Agriculture Science and Technology Development (Project No. 008494)" Rural Development Administration, Republic of Korea.
REFERENCES
- Abril, R., J. Garrett, S.G. Zeller, W.J. Sander and R.W. Mast, 2003. Safety assessment of DHA-rich microalgae from Schizochytrium sp. Part V: Target animal safety/toxicity study in growing swine. Regul. Toxicol. Pharmacol., 37: 73-82.
CrossRef - Bryhni, E.A., N.P. Kjos, R. Ofstad and M. Hunt, 2002. Polyunsaturated fat and fish oil in diets for growing-finishing pigs: Effects on fatty acid composition and meat, fat and sausage quality. J. Meat Sci., 62: 1-8.
CrossRefDirect Link - Calder, P.C., 1996. Immunomodulatory and anti-inflammatory effects of n-3 polyunsaturated fatty acids. Proc. Nutr. Soc., 55: 737-774.
CrossRef - Gardiner, G.E., A.J. Campbell, J.V. O'Doherty, E. Pierce and P.B. Lynch et al., 2008. Effect of Ascophyllum nodosum extract on growth performance, digestibility, carcass characteristics and selected intestinal microflora populations of grower-finisher pigs. Anim. Feed Sci. Technol., 141: 259-273.
CrossRefDirect Link - Coates, W. and R. Ayerza, 2009. Chia (Salvia hispanica L.) seed as an n-3 fatty acid source for finishing pigs: Effects on fatty acid composition and fat stability of the meat and internal fat, growth performance and meat sensory characteristics. J. Anim. Sci., 87: 3798-3804.
CrossRefDirect Link - Corino, C., S. Magni, E. Pagliarini, R. Rossi, G. Pastorelli and L.M. Chiesa, 2002. Effects of dietary fats on meat quality and sensory characteristics of heavy pig loins. Meat Sci., 60: 1-8.
CrossRef - Corino, C., M. Musella and J. Mourot, 2008. Influence of extruded linseed on growth, carcass composition and meat quality of slaughtered pigs at one hundred ten and one hundred sixty kilograms of liveweight. J. Anim. Sci., 86: 1850-1860.
CrossRefDirect Link - Dewailly, E., C. Blanchet, S. Lemieux, L. Sauve, S. Gingras, P. Ayotte and B.J. Holub, 2001. n-3 Fatty acids and cardiovascular disease risk factors among the Inuit of Nunavik. Am. J. Clin. Nutri., 74: 464-473.
Direct Link - Dyerberg, J., H.O. Bang and N. Hjorne, 1975. Fatty acid composition of the plasma lipids in Greenland Eskimos. Am. J. Clin. Nutr., 28: 958-966.
Direct Link - Edwards, R., M. Peet, J. Shay and D. Horrobin, 1998. Omega-3 polyunsaturated fatty acid levels in the diet and in red blood cell membranes of depressed patients. J. Affect. Disord., 48: 149-155.
CrossRef - Folch, J., M. Lees and G.H.S. Stanley, 1957. A simple method for the isolation and purification of total lipides from animal tissues. J. Biol. Chem., 226: 497-509.
CrossRefPubMedDirect Link - Goldberg, R.J. and J. Katz, 2007. A meta-analysis of the analgesic effects of omega-3 polyunsaturated fatty acid supplementation for inflammatory joint pain. Pain, 129: 210-223.
CrossRefPubMedDirect Link - He, M.L., W. Hollwich and W.A. Rambeck, 2002. Supplementation of algae to the diet of pigs: A new possibility to improve the iodine content in the meat. J. Anim. Physiol. Anim. Nutr., 86 : 97-104.
PubMed - Hintz, H.F., H. Heitman, W.C. Weir, D.T. Torell and J.H. Meyer, 1966. Nutritive value of algae grown on sewage. J. Anim. Sci., 25: 675-681.
Direct Link - Honikel, K.O., 1998. Reference methods for the assessment of physical characteristics of meat. Meat Sci., 49: 447-457.
CrossRefDirect Link - Irie, M. and M. Sakimoto, 1992. Fat characteristics of pigs fed fish oil containing eicosapentaenoic and docosahexaenoic acids. J. Anim. Sci., 70: 470-477.
Direct Link - Jaturasitha, S., Y. Wudthithumkanaporn, P. Rurksasen and M. Kreuzer, 2002. Enrichment of pork with omega-3 fatty acids by tuna oil supplements: Effects on performance as well as sensory, nutritional and processing properties of pork. Asian Aust. J. Anim. Sci., 15: 1622-1633.
Direct Link - Jaturasitha, S., R. Khiaosa-ard, P. Pongpiachan and M. Kreuzer, 2009. Early deposition of n-3 fatty acids from tuna oil in lean and adipose tissue of fattening pigs is mainly permanent. J. Anim. Sci., 87: 693-703.
CrossRefDirect Link - Jolly, C.A., Y.H. Jiang, R.S. Chapkin and D.N. McMurray, 1997. Dietary (n-3) polyunsaturated fatty acids suppress murine lymphoproliferation, interleukin-2 secretion, and the formation of diacylglycerol and ceramide. J. Nutr., 127: 37-43.
Direct Link - Kauffman, R.G., G. Eikelenboom, P.G. Van der Wal, B. Engel and M. Zaar, 1986. A comparison of methods to estimate waterholding capacity in post-rigor porcine muscle. Meat Sci., 18: 307-322.
CrossRef - Kouba, M., M. Enser, F.M. Whittington, G.R. Nute and J.D. Wood, 2003. Effect of a high-linolenic acid diet on lipogenic enzyme activities, fatty acid composition, and meat quality in the growing pig. J. Anim. Sci., 81: 1967-1979.
CrossRefDirect Link - Kruger, M.C. and D.F. Horrobin, 1997. Calcium metabolism, osteoporosis and essential fatty acids: A review. Prog. Lipid Res., 36: 131-151.
PubMed - Metcalfe, L.D., A.A. Schmitz and J.R. Pelka, 1966. Rapid preparation of fatty acid esters from lipids for gas chromatographic analysis. Anal. Chem., 38: 514-515.
CrossRefDirect Link - Piva, A., V. Pizzamiglio, M. Morlacchini, M. Tedeschi and G. Piva, 2007. Lipid microencapsulation allows slow release of organic acids and natural identical flavors along the swine intestine. J. Anim. Sci., 85: 486-493.
CrossRefPubMedDirect Link - Sardi, L., G. Martelli, L. Lambertini, P. Parisini and A. Mordenti, 2006. Effects of a dietary supplement of DHA-rich marine algae on Italian heavy pig production parameters. Livest. Sci., 103: 95-103.
CrossRefDirect Link - Turner, J.L., S.S. Dritz, J.J. Higgins and J.E. Minton, 2002. Effects of Ascophyllum nodosum extract on growth performance and immune function of young pigs challenged with Salmonella typhimurium. J. Anim. Sci., 80: 1947-1953.
Direct Link








