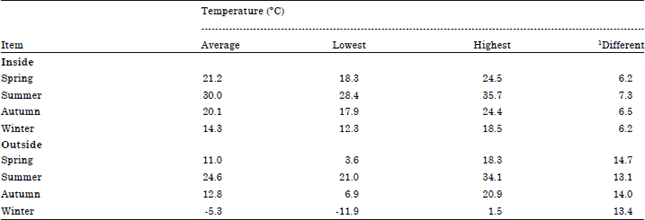
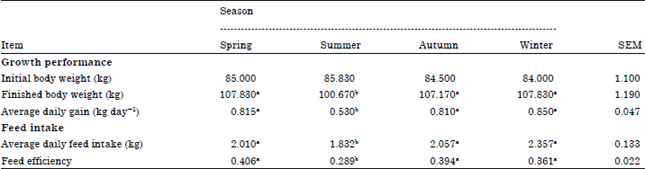
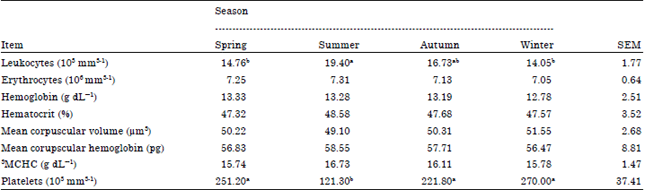
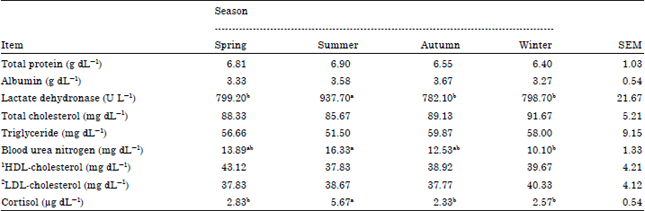
Research Article
Growth Performance, Blood Characteristics and Immune Responses of Fattening Pigs in Different Seasons
Swine Science and Technology Center, Gyeongnam National University of Science and Technology, Jinju, Korea
Young-Min Song
Department of Animal Resource Technology, Gyeongnam National University of Science and Technology, Jinju, Korea