Research Article
Genetic Diversity of Horseshoe Crab (Tachypleus gigas) in Malaysia Revealed using Microsatellite Markers
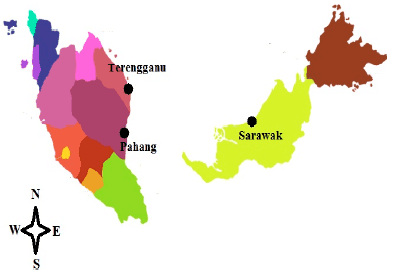
Department of Aquaculture, Faculty of Agriculture, Universiti Putra Malaysia, 43400 UPM, Serdang, Selangor, Malaysia
E. Ismail
School of Bioscience and Biotechnology, Faculty of Science and Technology, Universiti Kebangsaan Malaysia, 43600 UKM Bangi, Selangor D.E., Malaysia
B. Akbar John
Department of Biotechnology, Kulliyyah of Science, International Islamic University Malaysia, Jalan Sulthan Ahmed Shah, Bander Indera Mahkota, 25200, Kuantan, Pahang, Malaysia
K.C.A. Jalal
Department of Biotechnology, Kulliyyah of Science, International Islamic University Malaysia, Jalan Sulthan Ahmed Shah, Bander Indera Mahkota, 25200, Kuantan, Pahang, Malaysia
A. Mohd-Adnan
School of Bioscience and Biotechnology, Faculty of Science and Technology, Universiti Kebangsaan Malaysia, 43600 UKM Bangi, Selangor D.E., Malaysia