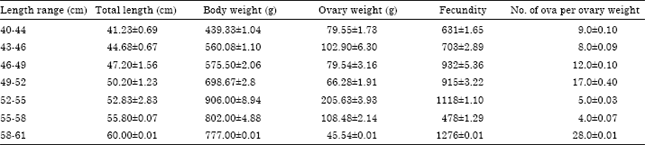
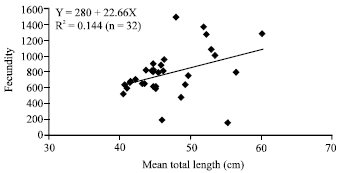
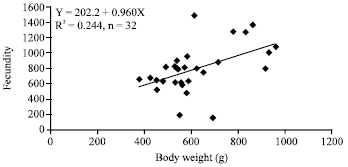
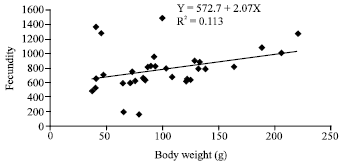
Research Article
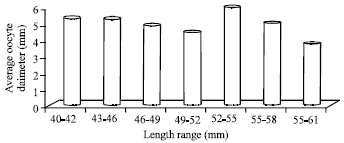
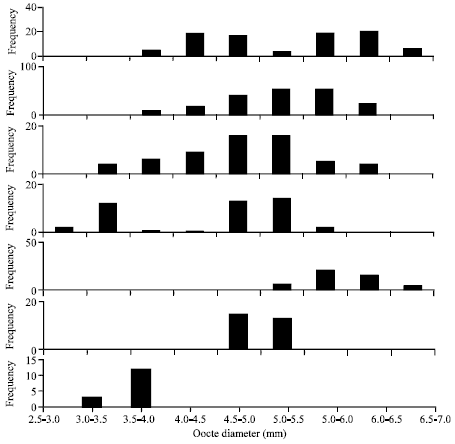
Fecundity and Egg Size of Grey-Eel Catfish Plotosus canius (Hamilton, 1822) from the Coastal Waters of Kampong Telok, Negeri Sembilan Peninsular Malaysia
Department of Aquaculture, Faculty of Agriculture, Universiti Putra Malaysia, 43400 Serdang, Selangor, Malaysia
S.M.N. Amin
Department of Aquaculture, Faculty of Agriculture, Universiti Putra Malaysia, 43400 Serdang, Selangor, Malaysia
A. Arshad
Department of Aquaculture, Faculty of Agriculture, Universiti Putra Malaysia, 43400 Serdang, Selangor, Malaysia
M. Aminur Rahman
Marine Biotechnology Laboratory, Institute of Bioscience, Universiti Putra Malaysia, Malaysia