Research Article
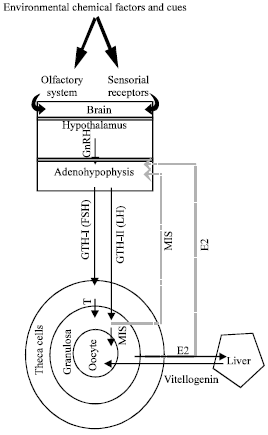
Endocrine Control of Oogenesis in Teleosts
Department of Aquaculture, Faculty of Agriculture, Universiti Putra Malaysia, 43400, Serdang, Selangor, Malaysia
Mohd Salleh Kamarudin
Department of Aquaculture, Faculty of Agriculture, Universiti Putra Malaysia, 43400, Serdang, Selangor, Malaysia
Sharr Azni Harmin
Center for Land and Aquatic Technology, Faculty of Science and Biotechnology, Universiti Selangor, 45600 Bestari Jaya, Selangor, Malaysia