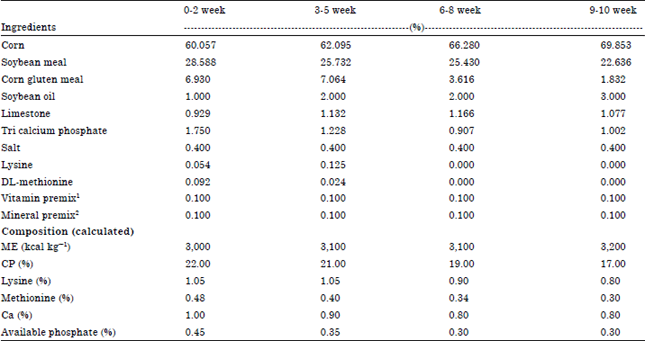
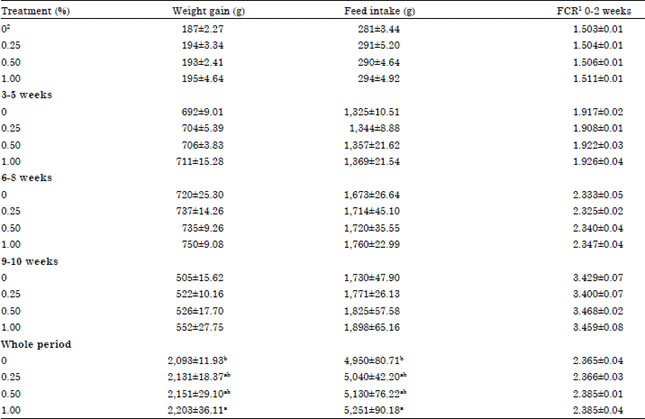
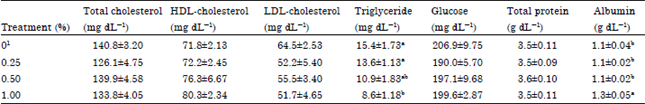
Research Article
Effect of Rubus coreanus Miquel Byproducts on Performance and Hormone Secretion of Crossbred Chicks
Department of Animal Science, Chonbuk National University, Jeonju 561-756, Korea
Y.D. Jeong
Department of Animal Science, Chonbuk National University, Jeonju 561-756, Korea
M.R. Hassan
Department of Animal Science, Chonbuk National University, Jeonju 561-756, Korea
C.W. Kang
College of Veterinary Medicine, Chonbuk National University Jeonju 561-756, Korea
Kyeong Seon Ryu
Department of Animal Science, Chonbuk National University, Jeonju 561-756, Korea