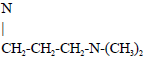Research Article
Differential Sensitivities of Three Strains of Drosophila melanogaster (Diptera: Drosophilidae) to Imipramine as an Antidepressant Drug
Department of Medical Entomology, School of Health and Nutrition, Research Centre for Health Sciences, Shiraz University of Medical Sciences, Shiraz, Iran
M.H. Eghbal
Shiraz Anesthesiology and Critical Care Research Centre, Medical School, Shiraz University of Medical Sciences, Shiraz, Iran
F. Eghbal
School of Medicine, Kazerun Branch, Islamic Azad University, Fars, Iran