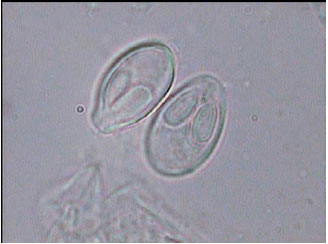
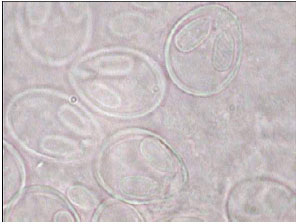
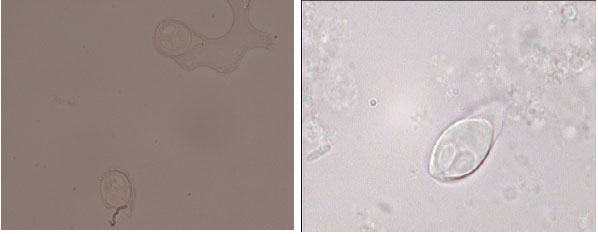
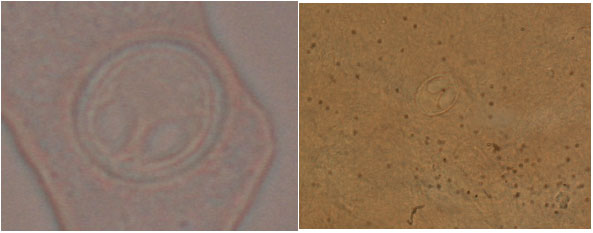
Research Article
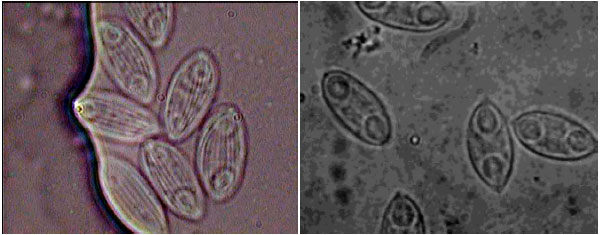
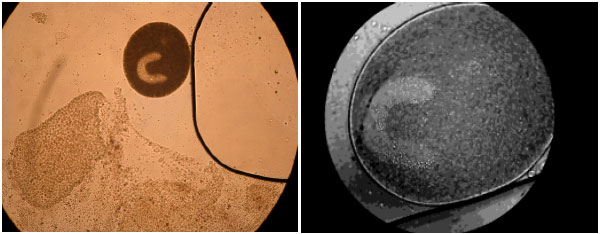
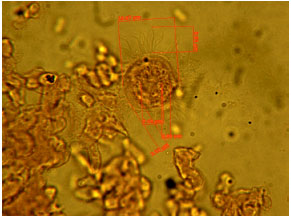
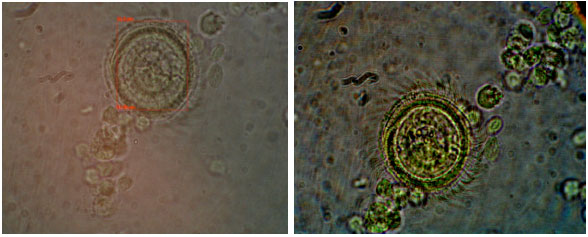
Protozoan and Myxozoan Infections in Some Fishes of Parishan Lake
Department of Aquatic Animal Health, Faculty of Veterinary Medicine, Kazerun Branch, Islamic Azad University, Kazerun, Iran
M. Masoumian
Department of Aquatic Animal Health, Fisheries Research Institute of Iran, Tehran, Iran
B. Jalali Jafari
Department of Aquatic Animal Health, Faculty of Specialized Veterinary Medicine, Islamic Azad University, Science and Research Branch, Tehran, Iran
M. Barzegar Dowlatabadi
MS in Fisheries Engineering, Tehran, Iran