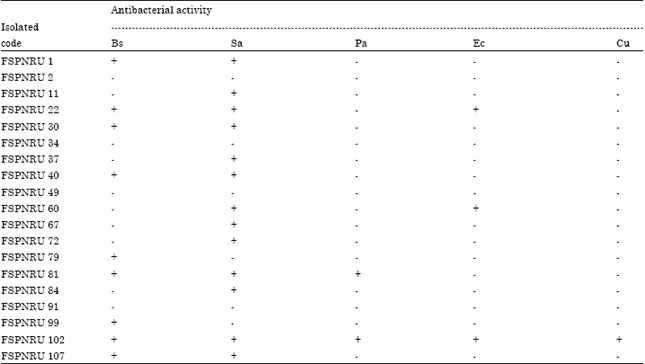
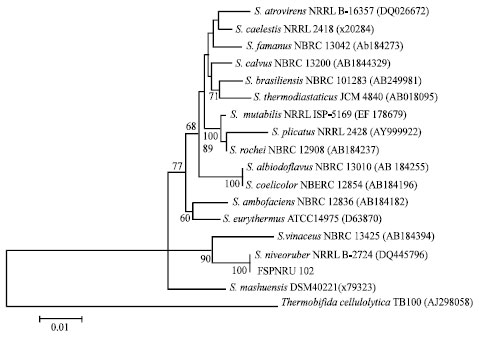
Research Article
Isolation and Identification of Actinomycetes from Termite’s Gut against Human Pathogen
Faculty of Science and Technology, Phranakhon Rajabhat University, Bangkok 10220, Thailand
N. Sripairoj
Faculty of Science and Technology, Phranakhon Rajabhat University, Bangkok 10220, Thailand
K. Sinma
Faculty of Science and Technology, Phranakhon Rajabhat University, Bangkok 10220, Thailand