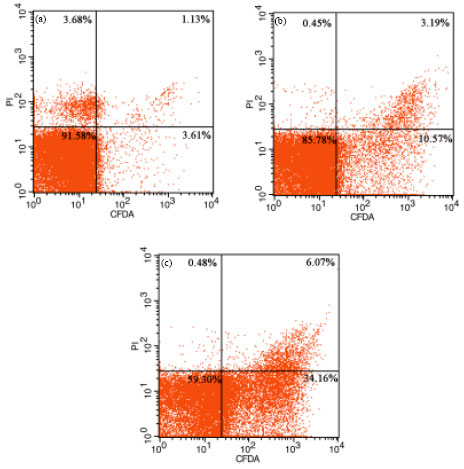
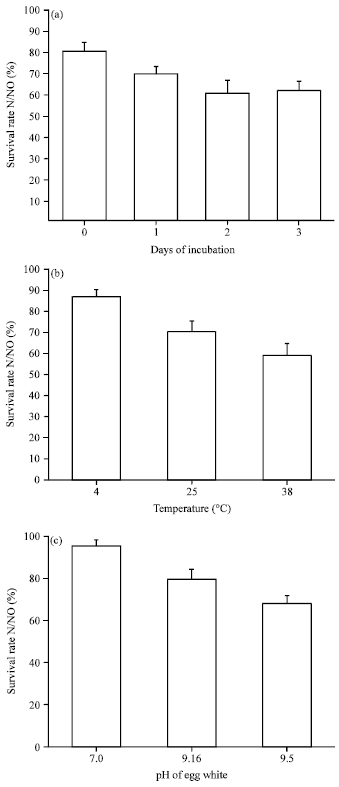
Research Article
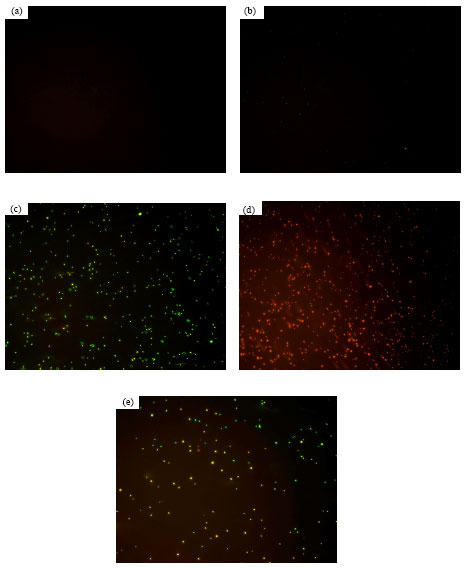
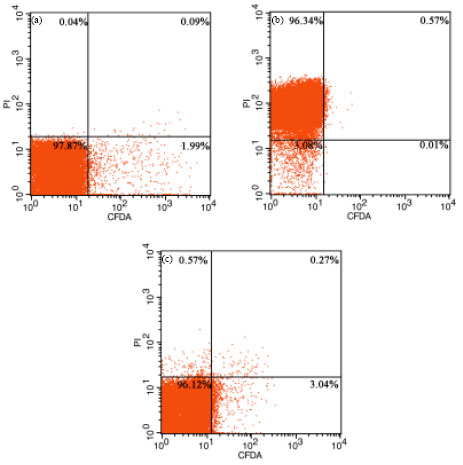
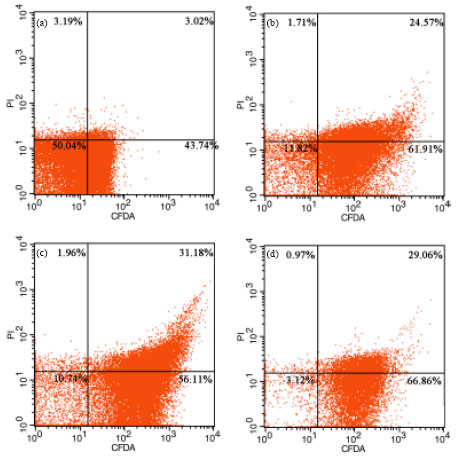
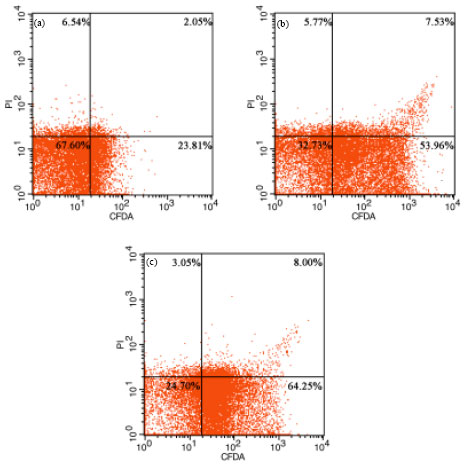
Assessment of Salmonella enteritidis Viability in Egg White during Early Incubation Stages by Fluorescent Staining Method
National R & D Center for Egg Processing, Huazhong Agricultural University, Wuhan, 430070, China
Meihu Ma
National R & D Center for Egg Processing, Huazhong Agricultural University, Wuhan, 430070, China
Yongguo Jin
National R & D Center for Egg Processing, Huazhong Agricultural University, Wuhan, 430070, China
Ning Qiu
National R & D Center for Egg Processing, Huazhong Agricultural University, Wuhan, 430070, China
Chan Wang
National R & D Center for Egg Processing, Huazhong Agricultural University, Wuhan, 430070, China
Guodong Ren
National R & D Center for Egg Processing, Huazhong Agricultural University, Wuhan, 430070, China
Xin Huang
National R & D Center for Egg Processing, Huazhong Agricultural University, Wuhan, 430070, China