Research Article
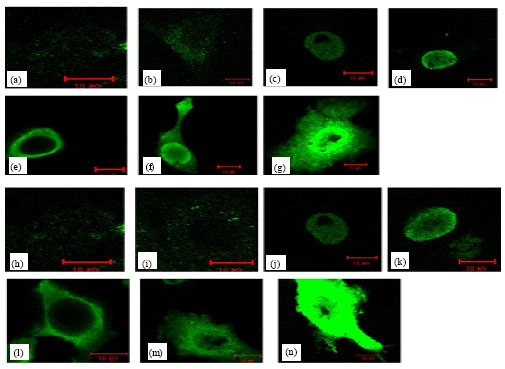
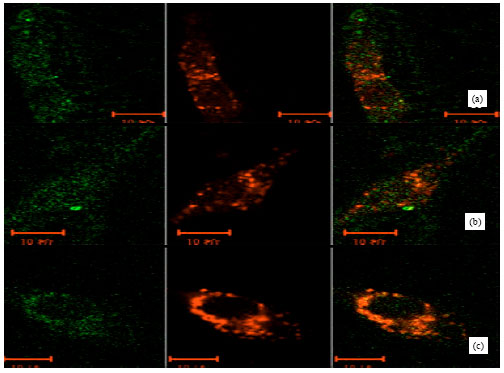
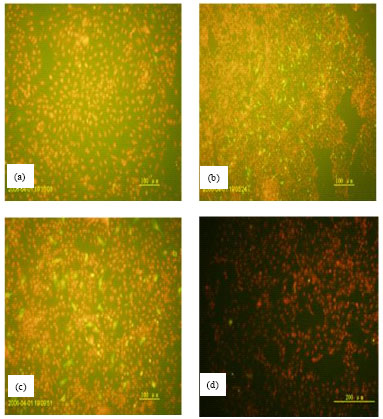
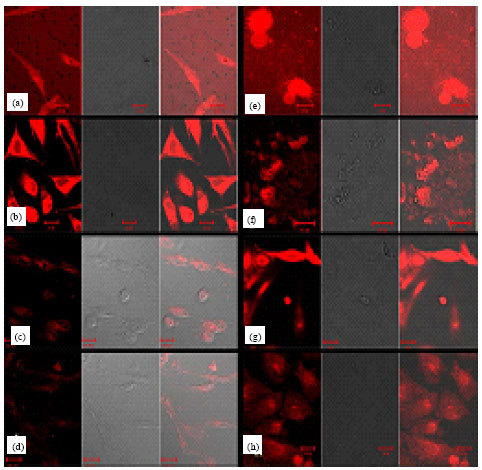
The Subcellular Localization and the Tissue Tropism of Canine Parvovirus based on the Co-localization of Transferrin Receptors
Hebei Key Laboratory of Preventive Veterinary, Department of Animal Science, Hebei Normal University of Science and Technology, Qinhuangdao 066600, China
Fei Zhong
Department of Basic Veterinary Medicine, College of Animal Science and Technology, Agricultural University of Hebei, Baoding 071001, China
Wangbin Cao
Hebei Key Laboratory of Preventive Veterinary, Department of Animal Science, Hebei Normal University of Science and Technology, Qinhuangdao 066600, China
Manfu Zhang
Laboratory for Animal Molecular Virology, College of Biological Sciences, China Agricultural University, Beijing 100094, China