Research Article
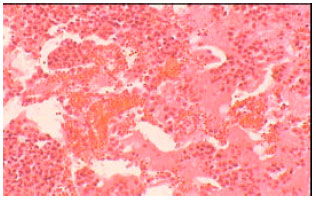
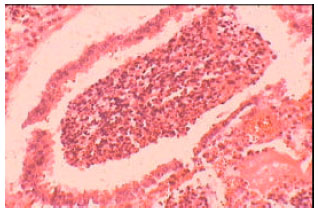
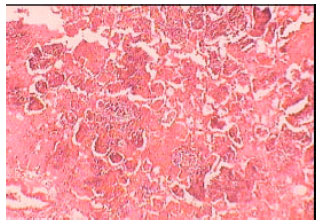
Isolation, Characterization and Antibiogram of Mycoplasma bovis in Sheep Pneumonia
Department of Veterinary Microbiology and Immunology,College of Veterinary Sciences and Animal Husbandry, Pt. Deen Dayal Uphaydhay Pashu Chiktsa Vigyan Vishvidyalaya Wvum Go anusandhan Sansthan (DUVASU), Mathura-281001, India
Amit K. Verma
Department of Veterinary Epidemiology and Preventive Medicine, College of Veterinary Sciences and Animal Husbandry, Pt. Deen Dayal Uphaydhay Pashu Chiktsa Vigyan Vishvidyalaya Wvum Go anusandhan Sansthan (DUVASU), Mathura-281001, India
Neeraj K. Gangwar
Department of Veterinary Pathology, College of Veterinary Sciences and Animal Husbandry, Pt. Deen Dayal Uphaydhay Pashu Chiktsa Vigyan Vishvidyalaya Wvum Go anusandhan Sansthan (DUVASU), Mathura-281001, India
Anu Rahal
Department of Veterinary Pharmacology and Toxicology, College of Veterinary Sciences and Animal Husbandry, Pt. Deen Dayal Uphaydhay Pashu Chiktsa Vigyan Vishvidyalaya Wvum Go anusandhan Sansthan (DUVASU), Mathura-281001, India