Research Article
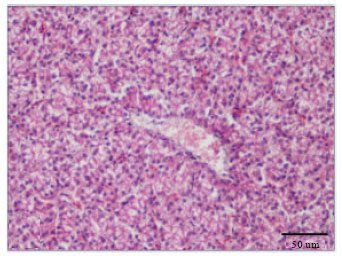
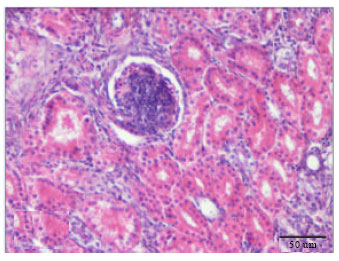
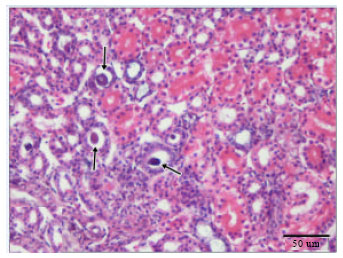
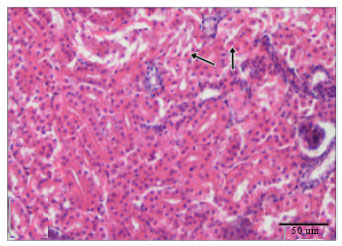
Cadmium Induced Changes on Growth Performance, Some Biochemical Parameters and Tissue in Broilers: Effects of Vitamin C and Vitamin E
Department of Biochemistry,Faculty of Veterinary Medicine, Kirikkale University, Yahsihan, 71451 Kirikkale, Turkey
A. Arzu Yigit
Department of Physiology, Faculty of Veterinary Medicine, Kirikkale University, Yahsihan, 71451 Kirikkale, Turkey
Ilkay Yalcinkaya
Department of Animal Nutrition and Nutritional Diseases, Faculty of Veterinary Medicine, Kirikkale University, Yahsihan, 71451 Kirikkale, Turkey
Ertan Oruc
Department of Pathology, Faculty of Veterinary Medicine, Ataturk University, 25240, Erzurum, Turkey
Ozkan Duru
Department of Biochemistry,Faculty of Veterinary Medicine, Kirikkale University, Yahsihan, 71451 Kirikkale, Turkey
Metin Arslan
Department of Chemistry and Chemical Processing Technologies, Kirikkale Vocational High School, Kirikkale University, 71451 Kirikklale, Turkey