Research Article
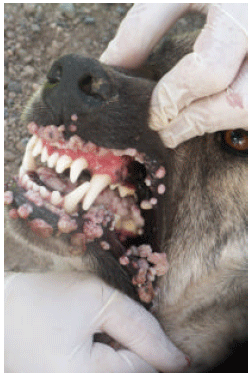
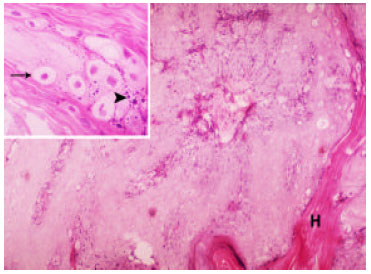
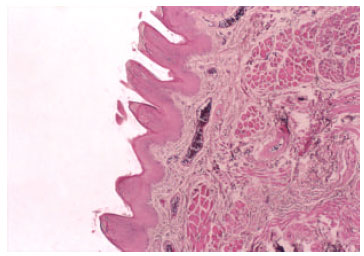
The Efficacy of Tarantula cubensis Extract (Theranekron) in Treatment of Canine Oral Papillomatosis
Department of Internal Medicine, Faculty of Veterinary Medicine, University of Dicle, 21180-Diyarbakir, Turkey
S. Sekin
Department of Internal Medicine, Faculty of Veterinary Medicine, University of Dicle, 21180-Diyarbakir, Turkey
A. Simsek
Department of Internal Medicine, Faculty of Veterinary Medicine, University of Dicle, 21180-Diyarbakir, Turkey
A. Kochan
Department of Internal Medicine, Faculty of Veterinary Medicine, University of Dicle, 21180-Diyarbakir, Turkey
S. Tunik
Department of Histology and Embryology, Faculty of Medicine, University of Dicle, Diyarbakir, Turkey