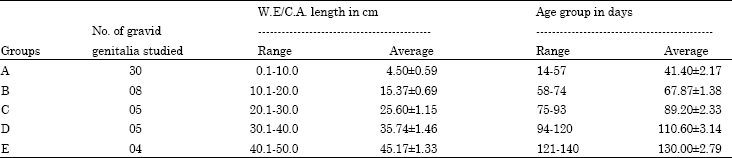
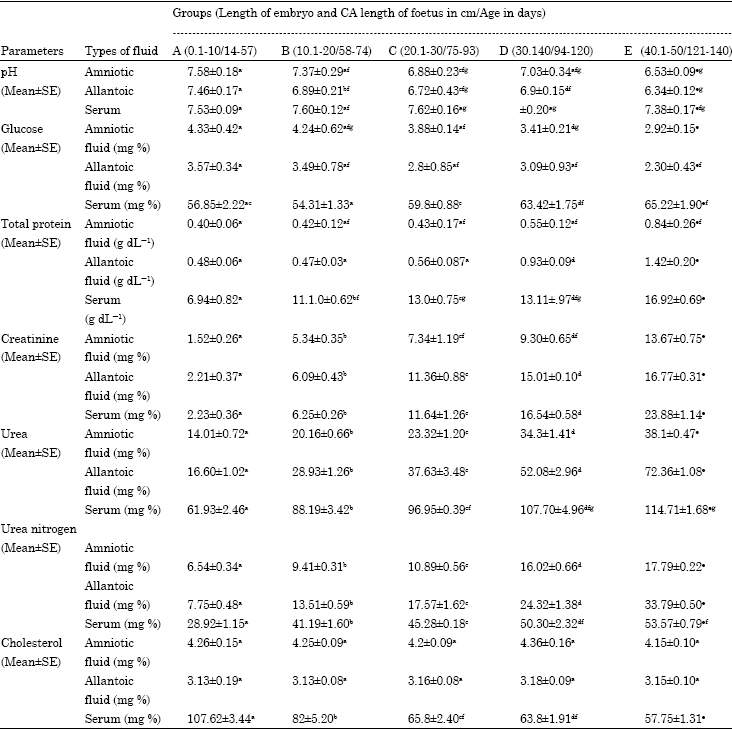
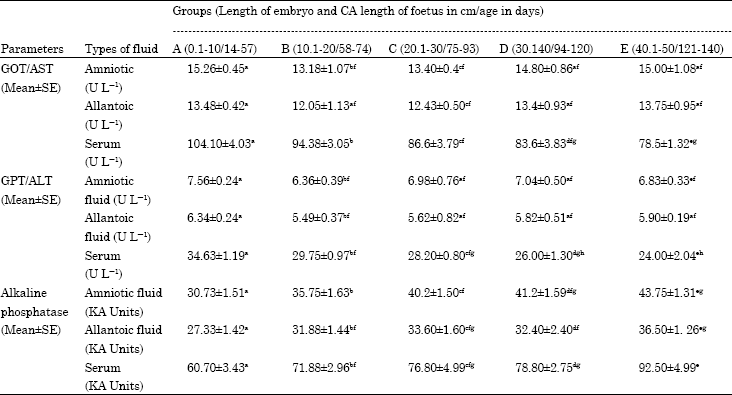
Research Article
Biochemical Indices in Sheep During Different Stages of Pregnancy
Division of Animal Reproduction, Gynaecology and Obstetrics,Faculty of Veterinary Sciences and Animal Husbandry,S.K. University of Agricultural Sciences and Technology, Shuhama,Alusteng, Srinagar-190006, Jammu Kashmir, India
G.M. Wani
Division of Animal Reproduction, Gynaecology and Obstetrics,Faculty of Veterinary Sciences and Animal Husbandry,S.K. University of Agricultural Sciences and Technology, Shuhama,Alusteng, Srinagar-190006, Jammu Kashmir, India
J.I.A. Bhat
Division of Animal Reproduction, Gynaecology and Obstetrics,Faculty of Veterinary Sciences and Animal Husbandry,S.K. University of Agricultural Sciences and Technology, Shuhama,Alusteng, Srinagar-190006, Jammu Kashmir, India
A.R. Choudhury
Division of Animal Reproduction, Gynaecology and Obstetrics,Faculty of Veterinary Sciences and Animal Husbandry,S.K. University of Agricultural Sciences and Technology, Shuhama,Alusteng, Srinagar-190006, Jammu Kashmir, India
M.Z. Khan
Division of Animal Reproduction, Gynaecology and Obstetrics,Faculty of Veterinary Sciences and Animal Husbandry,S.K. University of Agricultural Sciences and Technology, Shuhama,Alusteng, Srinagar-190006, Jammu Kashmir, India