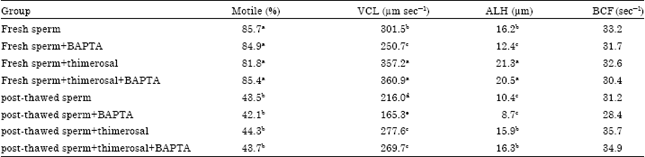
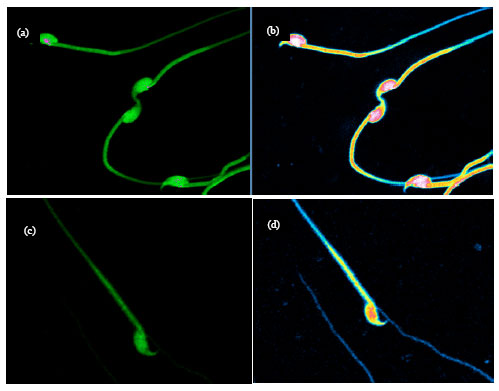
Research Article
Impact on Hyperactivated Motility of Cryopreserved Mouse Sperm from Pretreatment with Thimerosal
Laboratory of Animal Embryonic Biotechnology, College of Animal Science and Technology and State Key Laboratories for Agrobiotechnology, China Agricultural University, Beijing, 100193, Peoples� Republic of China
Guang-Bin Zhou
Institute of Animal Genetics and Breeding, College of Animal Science and Technology, Sichuan Agricultural University (Chengdu Campus), Wenjiang, 611130, Peoples� Republic of China
Yan Zeng
Laboratory of Animal Embryonic Biotechnology, College of Animal Science and Technology and State Key Laboratories for Agrobiotechnology, China Agricultural University, Beijing, 100193, Peoples� Republic of China
Jun-Jie Li
Laboratory of Animal Embryonic Biotechnology, College of Animal Science and Technology and State Key Laboratories for Agrobiotechnology, China Agricultural University, Beijing, 100193, Peoples� Republic of China
Qing-Jing Zhang
Laboratory of Animal Embryonic Biotechnology, College of Animal Science and Technology and State Key Laboratories for Agrobiotechnology, China Agricultural University, Beijing, 100193, Peoples� Republic of China
Yun-Peng Hou
Laboratory of Animal Embryonic Biotechnology, College of Animal Science and Technology and State Key Laboratories for Agrobiotechnology, China Agricultural University, Beijing, 100193, Peoples� Republic of China
Shi-En Zhu
Laboratory of Animal Embryonic Biotechnology, College of Animal Science and Technology and State Key Laboratories for Agrobiotechnology, China Agricultural University, Beijing, 100193, Peoples� Republic of China