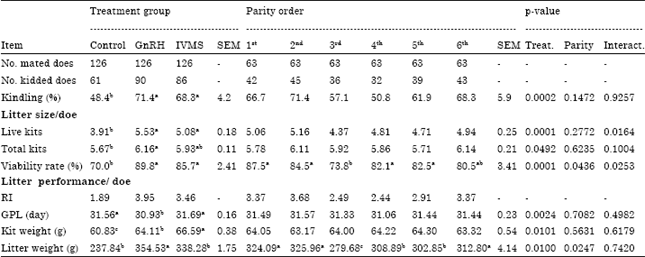
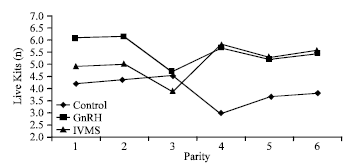
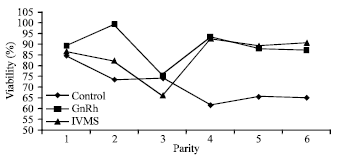
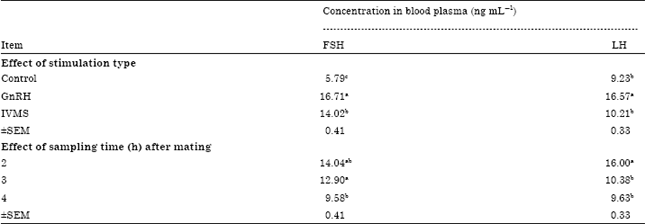
Research Article
Effect of Repeated GnRH Injection and Intra-vaginal Mechanical Stimulation on Reproductive Performance of Doe Rabbits
Department of Poultry Production, Faculty of Agriculture, Mansoura University, Mansoura, Egypt
A.E. Abdel-Khalek
Department of Animal Production, Faculty of Agriculture, Mansoura University, Mansoura, Egypt