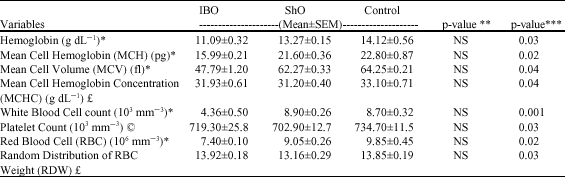
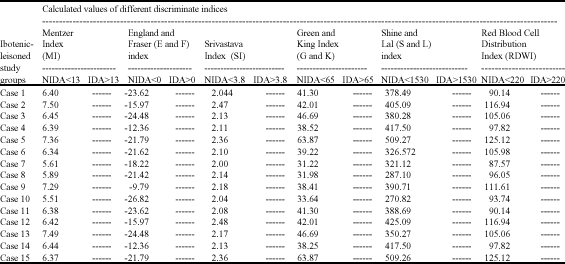
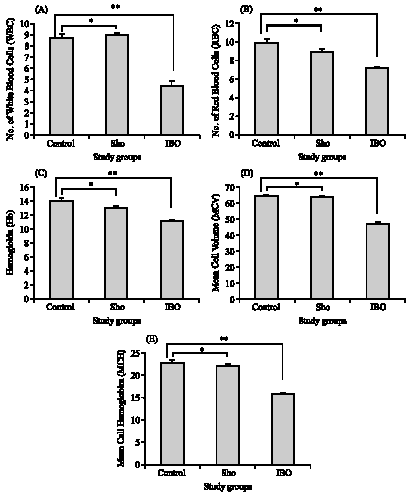
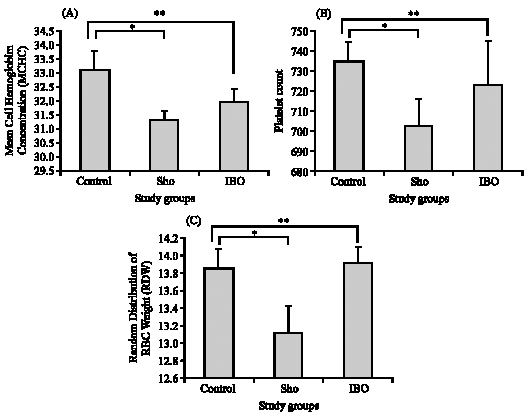
Research Article
Ibotenic Acid-Induced Hematological Disorders in Rat Model of Alzheimer's Disease
Physiology Research Center, Ahwaz Jondishapour University of Medical Sciences, Ahwaz, Iran
B. Keikhaei
Research Center of Thalassemia and Hemoglobinopathies, Shafa Hospital, Ahwaz Jondishapour University of Medical Sciences, Ahwaz, Iran
A. Sarkaki
Physiology Research Center, Ahwaz Jondishapour University of Medical Sciences, Ahwaz, Iran
A.H. Doulah
Department of Nursing, Ahwaz Azad University, Ahwaz, Iran