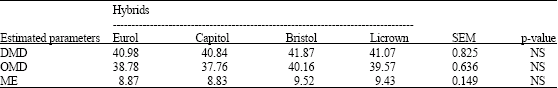
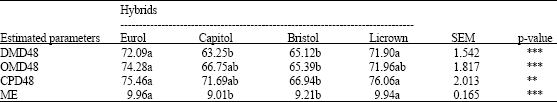
Research Article
In situ Rumen Degradability, in vitro Digestibility and in vitro Gas Production of Full Fat Canola Seeds
Department of Animal Science, Faculty of Agriculture, University of Ondokuz Mayis, Samsun, Turkiye
A.V. Garipoglu
Department of Animal Science, Faculty of Agriculture, University of Ondokuz Mayis, Samsun, Turkiye