Research Article
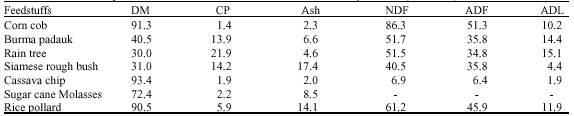
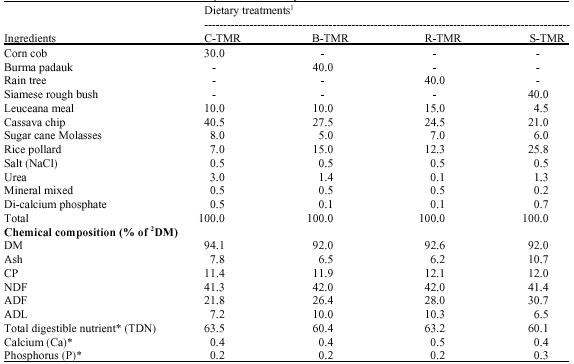
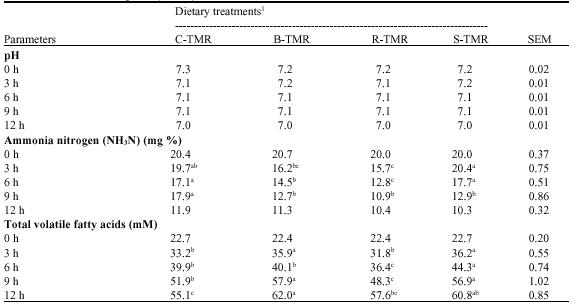
Effect of Burma Padauk (Plerocarpus indicus), Rain Tree (Samanea saman (Jacg.) Merr.) and Siamese Rough Bush (Streblus asper) Leaves as Fiber Sources in Total Mixed Ration on in vitro Fermentation
Animal Feed Resource and Animal Nutrition Research Unit, Division of Animal Science, Faculty of Veterinary Medicine and Animal Sciences, Mahasarakham University, Mahasarakham 44000, Thailand
O. Pimpa
Faculty of Technology and Management, Division of Bioprocessing, Prince of Songkla University Surat Thani Campus, Thailand










dr.nomitha das gupta Reply
im doing rescherch onthe antimicrobial affect of samanea saman on bacteria.if u can help me try to send me paper which i need in future