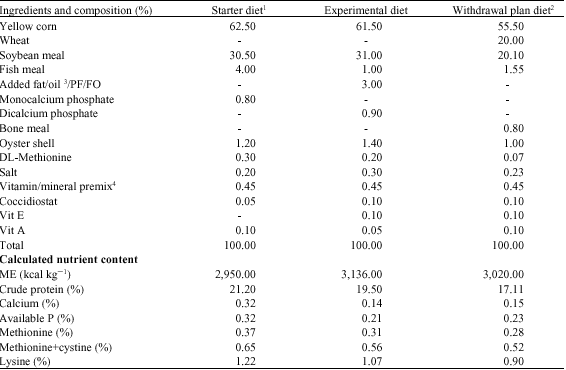
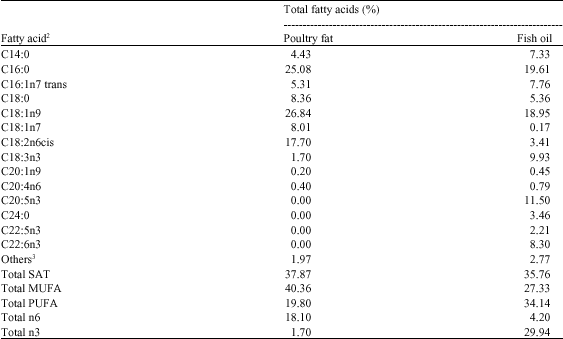
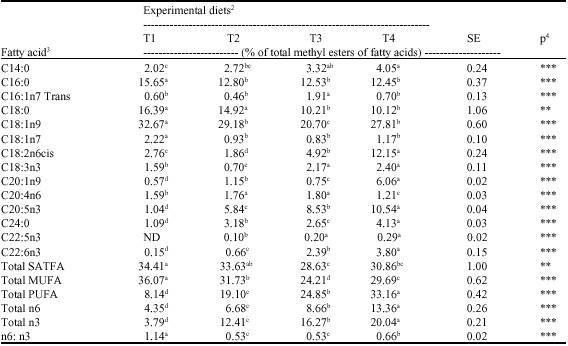
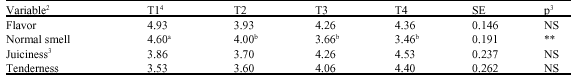
Research Article
Omega-3 Fatty Acids Enrichment and Organoleptic Characteristics of Broiler Meat
Department of Animal Science, Islamic Azad University,Shabestar Branch, Shabestar, Iran
Habib Aghdam Shahriar
Department of Animal Science, Islamic Azad University,Shabestar Branch, Shabestar, Iran
Naser Maheri-Sis
Department of Animal Science, Islamic Azad University,Shabestar Branch, Shabestar, Iran
Ali-Reza Ahmadzadeh
Department of Animal Science, Islamic Azad University,Shabestar Branch, Shabestar, Iran
Tohid Vahdatpoor
Department of Animal Science, Islamic Azad University,Shabestar Branch, Shabestar, Iran