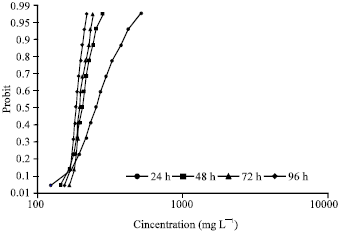
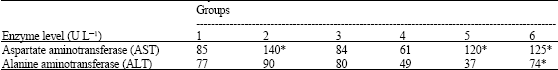
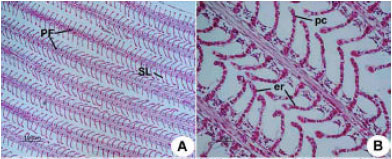
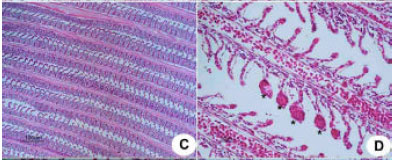
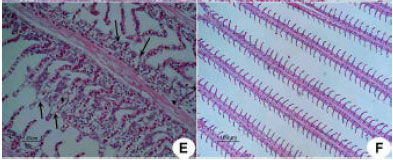
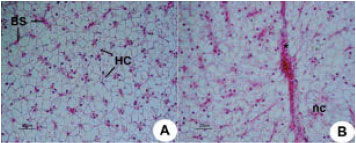
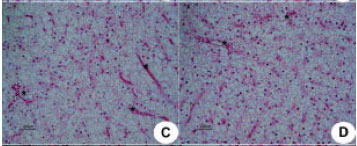
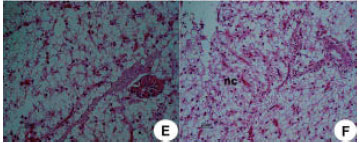
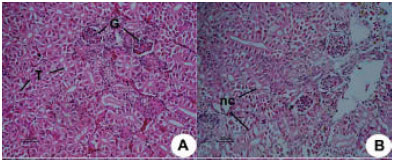
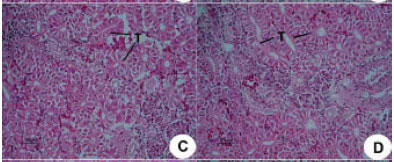
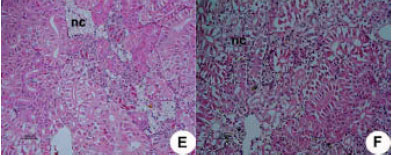
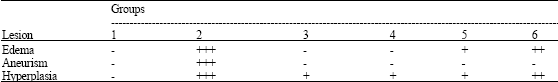
Research Article
Dietary Calcium Reducing Effects of Waterborne Lead Uptake in Nile Tilapia (Oreochromis niloticus)
Department of Pathobiology, Faculty of Science, Mahidol University, Bangkok 10400, Thailand
Wannee Jiraungkoorskul
Department of Pathobiology, Faculty of Science, Mahidol University, Bangkok 10400, Thailand
Somphong Sahaphong
Department of Pathobiology, Faculty of Science, Mahidol University, Bangkok 10400, Thailand
Tawewan Tansatit
Faculty of Veterinary Medicine, Mahidol University, Thailand
Piya Kosai
Department of Pathobiology, Faculty of Science, Mahidol University, Bangkok 10400, Thailand
Raviporn Madarasmi
Mahidol University International College, Mahidol University, Salaya Campus, Nakhonpathom 73170, Thailand