Research Article
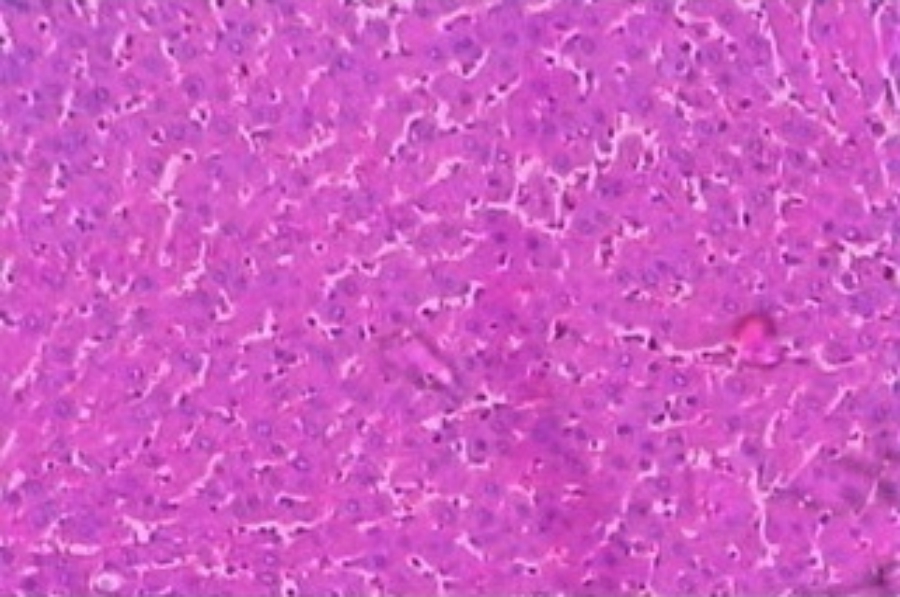
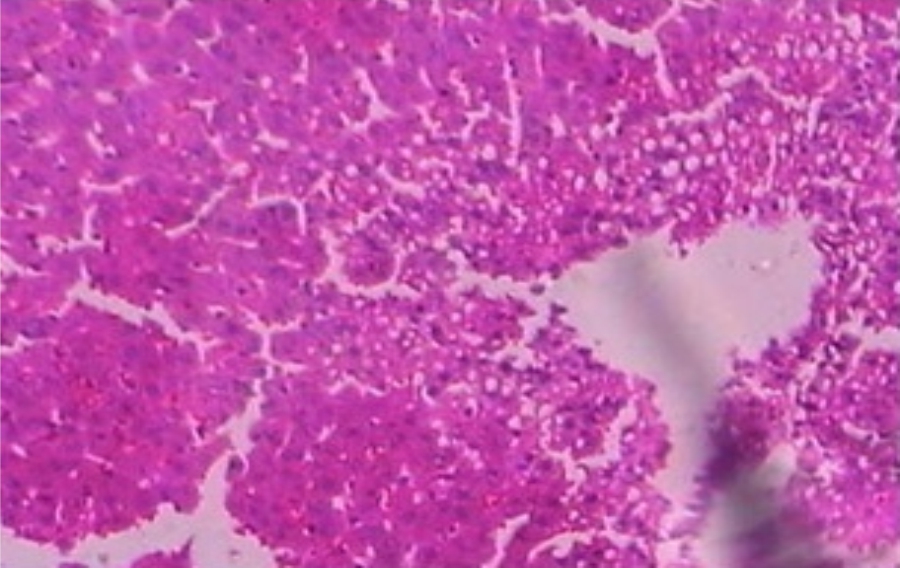
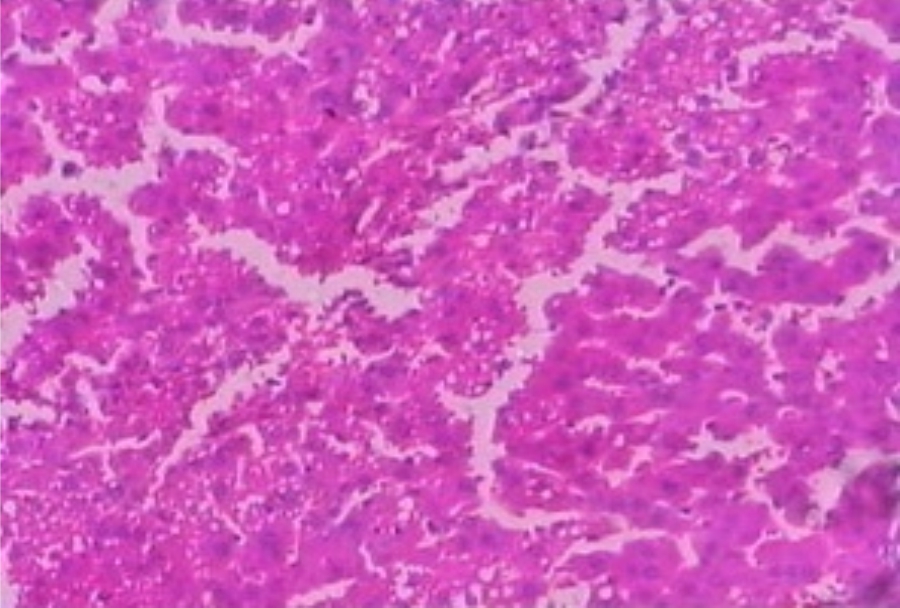
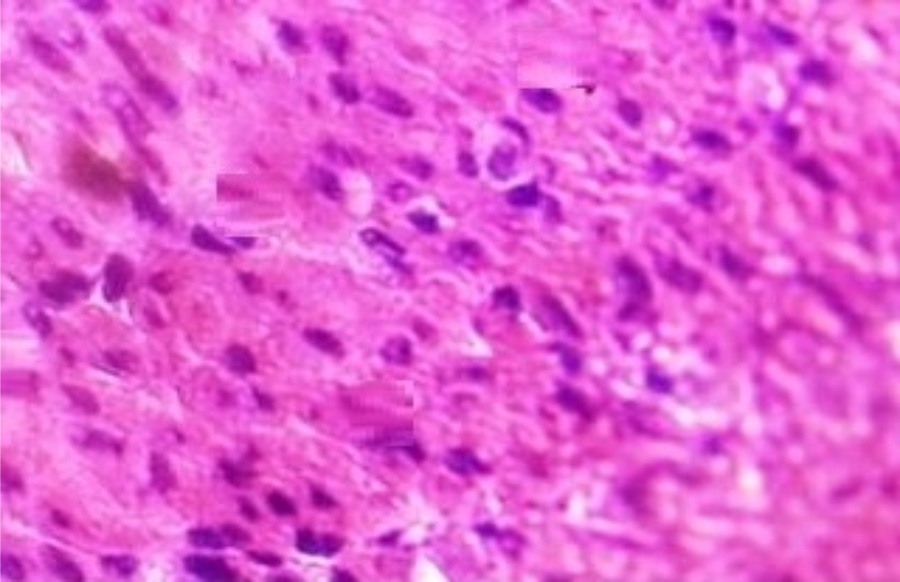
Hepatoprotective Activity of Martynia annua L. Leaves Against CCl4-induced Hepatic Damage in Rats
Guru Gobind Singh College of Pharmacy, Yamuna Nagar, Haryana, India
LiveDNA: 91.17253
Bhawna Chopra
Guru Gobind Singh College of Pharmacy, Yamuna Nagar, Haryana, India