Research Article
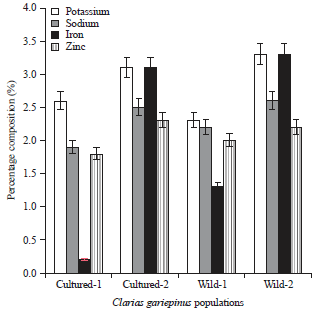
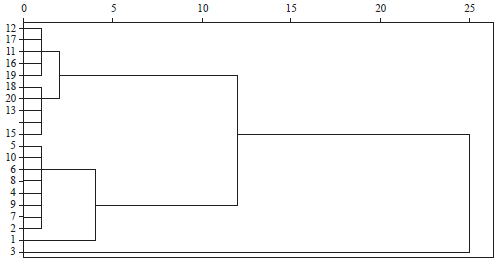
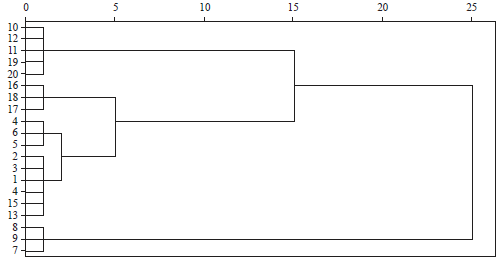
Genetic Relatedness of Clarias gariepinus (L.) from Cultured and Wild Populations Using Multivariate Analyses
Department of Genetics and Biotechnology, University of Calabar, PMB1115, Calabar, Nigeria
O.U. Udensi
Department of Genetics and Biotechnology, University of Calabar, PMB1115, Calabar, Nigeria
LiveDNA: 234.3756
M.C. Okolo
Department of Genetics and Biotechnology, University of Calabar, PMB1115, Calabar, Nigeria
F.U. Ogban
Department of Computer Science, University of Calabar, PMB1115, Calabar, Nigeria
N.G. Ufford
Department of Genetics and Biotechnology, University of Calabar, PMB1115, Calabar, Nigeria
E.U. Odo
Department of Genetics and Biotechnology, University of Calabar, PMB1115, Calabar, Nigeria
B.O. Asuquo
Department of Animal Science, University of Calabar, PMB1115, Calabar, Nigeria