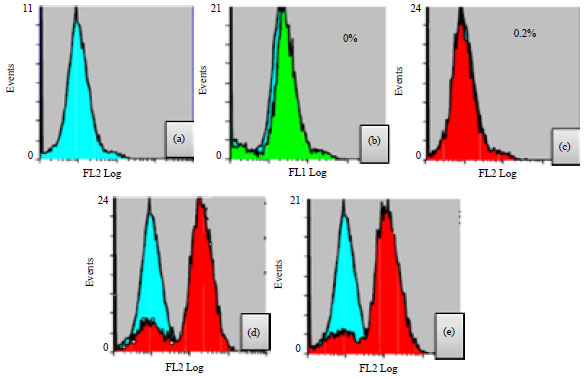
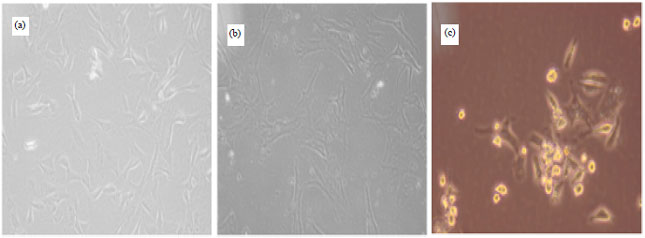
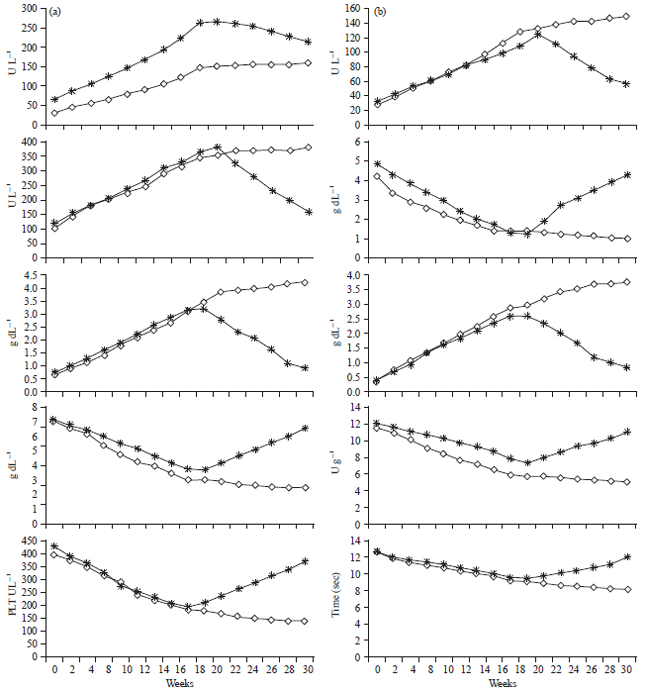
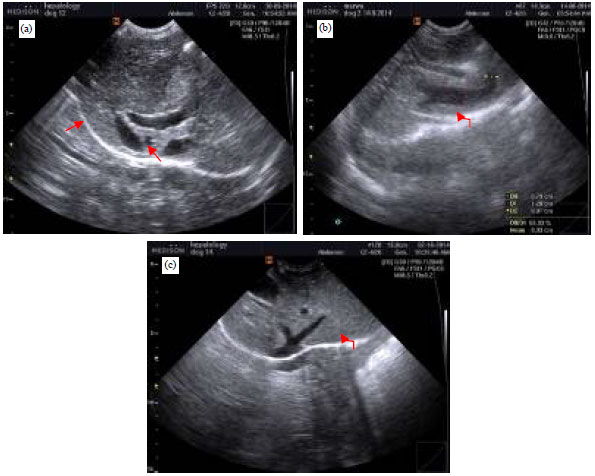
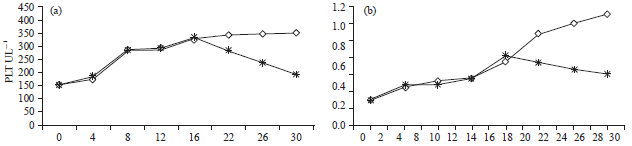
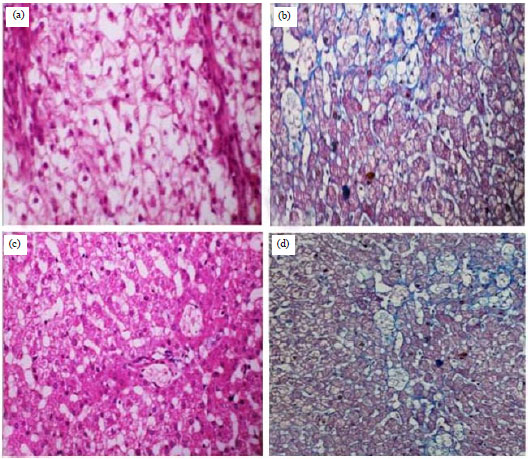
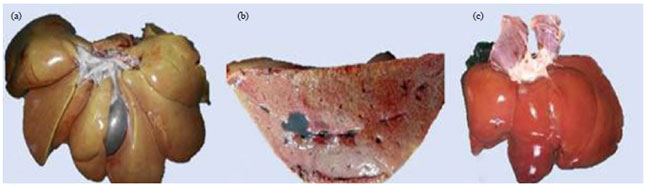
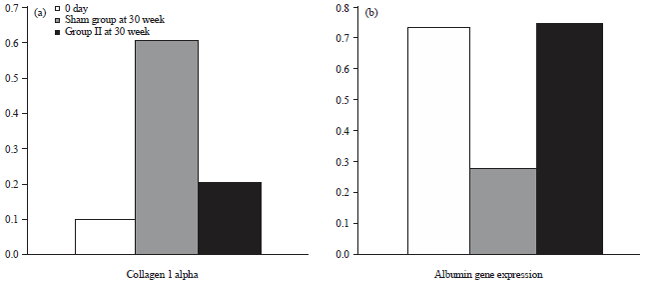
Research Article
Role of Hepatocyte Differentiated Mesenchymal Stem Cells in Treatment of Experimentally Induced Canine Liver Cirrhosis
Department of Surgery, Anesthesiology and Radiology, Faculty of Veterinary Medicine, Cairo University, Egypt
Ashraf A. Shamaa
Department of Surgery, Anesthesiology and Radiology, Faculty of Veterinary Medicine, Cairo University, Egypt
LiveDNA: 20.12761
Elham A. Hassan
Department of Surgery, Anesthesiology and Radiology, Faculty of Veterinary Medicine, Cairo University, Egypt
Omar S. El-Tookhy
Department of Surgery, Anesthesiology and Radiology, Faculty of Veterinary Medicine, Cairo University, Egypt
Sabry D. Abd Elfatah
Department of Medical Biochemistry and Molecular Biology, Faculty of Medicine, Cairo University, Egypt
Gehan G. Shehab
Department of Pathology, Animal Health Research Institute, Ministry of Agriculture, Egypt