Review Article
Exogenous Enzymes in Ruminant Nutrition: A Review
Faculty of Agriculture, University of Ruhuna, Sri Lanka
R.T. Seresinhe
Faculty of Agriculture, University of Ruhuna, Sri Lanka
Worldwide demand for animal based products are increasing in a booming rate thus emphasizing the essentiality of applying strategies to improve animal productivity. The major constraints in today’s livestock sector are high feed cost and low quality of available feed resources, especially in tropical developing countries. Over the years animal nutritionists have developed various physical, chemical and biological methods to overcome the problems associated with livestock feed stuffs. With the emerging concerns of food safety issues related with animal based products, application of biological treatment methods are in the center of attraction. As a biological treatment method utilization of exogenous enzymes has attracted a growing attention of researches and it has become a widely discussed theme among animal nutritionists (McAllister et al., 2003). Use of exogenous enzymes in monogastric animal diets (Bedford, 2000) is not a novel trend though the enzyme supplementation in ruminant diets is yet to be established in practical application. Early research attempts on supplementation ruminant diet with exogenous enzymes has a considerable long history which took place in 1960’s, mainly considering the amylolytic (McCarthy et al., 2013) and proteolytic (Vera et al., 2012) enzymes. But the inconsistency of the obtained results, less knowledge on mode of action of enzymes and the high production cost of enzymes hindered the further development of this field. With the recent advances in biotechnology, the reduced production cost of enzymes and better defined commercial enzyme products have prompted researchers to revise the potential of exogenous enzymes to improve the feed utilization in ruminants. Recently conducted several researches have demonstrated the beneficial effects of supplementing ruminant diets with exogenous enzymes more specifically, exogenous fibrolytic enzymes in means of improving fiber digestibility and consequently, the efficiency of feed utilization by ruminants in in vitro (Rodrigues et al., 2008; Murad et al., 2009; Azzaz, 2009), in vivo (Jalilvand et al., 2008; Krueger et al., 2008; Arriola et al., 2011) and in situ. Meanwhile some other studies (Elwakeel et al., 2007; Miller et al., 2008) reported no significant differences in digestibility and performance parameters with enzyme supplementation. With the immense effort of researchers the use of exogenous enzymes to enhance quality and digestibility of ruminant feeds is on the verge of delivering practical benefits, even though there are some issues such as mode of action of enzymes, synergism between exogenous enzymes and rumen microflora, suitable application method and optimum dosage to be spell out in a more precise manner.
The purpose of this review is to provide the summarized overview about aspects related with exogenous enzyme utilization in ruminant diets up to present time and to converse the future research directions.
TYPES, SOURCES AND EXTRACTION OF ENZYMES
Exogenous enzymes which use in ruminant nutrition can be characterized into three main categories as fibrolytic, amylolytic and proteolytic enzymes. In additional to major categories of enzymes, phytase which is extensively used in monogastric feeding is also becoming popular in ruminant feeding. Supplementing fibrolytic enzymes with ruminant diets has been the research interest in many studies (Yang et al., 2011; Chung et al., 2012; Mohamed et al., 2013) as digestion of fiber fraction in ruminant’s digestive system only reaches to the 65-70% even under ideal conditions. Exogenous fibrolytic enzymes can be classified further based on their specific activity as cellulase, which hydrolyze the fiber of plant cell wall to glucose, cellobiose or cellooligosaccharides with combined activity of three enzymes namely endoglucanase, exoglucanases and β-glucosidase (Zhang and Lynd, 2004). Xylanase, that catalyzes the hydrolysis of 1,4-beta-D-xylosidic linkages in xylans that are constituents of hemicellulose, a structural component of plant cell walls. Xylanolytic enzyme group includes endo-β-1,4-xylanase (1,4-β-D xylan xylanohydrolase, EC 3.2.1.8), which attack the main chain of xylans and β-D-xylosidase (1,4-β-xylan xylanohydrolase, EC 3.2.1.37), which hydrolyze xylooligosaccharides into D-xylose, in addition to a variety of debranching enzymes that is, α-L-arabino-furanosidases, α-glucuronidases and acetyl esterases (Kamble and Jadhav, 2012). Though researches hypothesized that the digestion of starch and protein is not limited within rumen, recent researches conducted with enzyme supplementation with amylolytic activity (Noziere et al., 2014; Klingerman et al., 2009; Gencoglu et al., 2010) and proteolytic activity (Eun and Beauchemin, 2005; Vera et al., 2012) have examined the potential of these enzymes to improve animal performance. Amylase has been divided into three sub classes as α, β and γ amylase based on the bonding type (Gurung et al., 2013). Knowlton et al. (2007) and Kincaid et al. (2005) conducted researches supplementing exogenous phytase enzyme in dairy cow rations with the primary focus of disclosing its effect on phosphorous digestibility.
| Table 1: | Cellulase and xylanase producing microorganisms and optimum conditions for the production (Motta et al., 2013; Sadhu and Maiti, 2013) (modified) |
 | |
Enzyme products are derived primarily from four bacterial (Bacillus subtilis, Lactobacillus acidophilus, L. plantarum and Streptococcus faecium, spp.), three fungal (Aspergillus oryzae, Trichoderma reesei and Saccharomyces cerevisiae) species and some yeasts (Table 1).
Cellulase are produced using both fungi and bacteria with more emphasis on the use of fungi because of their capability to produce ample amounts of enzymes (Subramaniyam and Vimala, 2012) and often less complex than bacterial cellulase and easy for extraction and purification. However, with the refining knowledge on microbiology the isolation and characterization of novel cellulase from bacteria are now becoming more popular. Behind this high acceptance there are several reasons like (1) Bacteria often have a higher growth rate than fungi allowing for higher recombinant production of enzymes, (2) Bacterial cellulases are often more complex and are in multi-enzyme complexes providing increased function and synergy and (3) Bacteria inhabit a wide variety of environmental and industrial niches like thermophilic or psychrophilic, alkaliphilic or acidophilic and halophilic strains (Immanuel et al., 2006). In another study rice filed soil was successfully utilized to isolate nine efficient cellulolytic microorganisms and produce cellulase enzymes mainly extracellularly (Lo et al., 2009).
Bacteria, yeasts and filamentous fungi have been identified as suitable candidates to produce xylanases (Kamble and Jadhav, 2012). Some of the most important xylanolytic enzyme producers include Aspergillus, Trichoderma, Streptomyces, Phanerochaetes, Chytridiomycetes, Ruminococcus, Fibrobacteres, Clostridia and Bacillus (Cai et al., 2004) (Table 1). Kuhad et al. (2006) reported extraction of xylanase producing bacteria from soil while significant amount of xylanase amount was extracted from a new isolate of Bacillus pumilus (Kumar et al., 2010). In a very recent study new xylanase producing Gram positive bacteria has isolated from termite gut (Matteotti et al., 2012).
Aspergillus oryzae is the major microorganism used to extract amylase enzyme (Tricarico et al., 2008) while in another study they have used amylase extracted from Bacillus licheniformis (Rojo et al., 2005). Apart from these microorganisms Chromohalobacter sp., Halobacillus sp., Haloarcula hispanica, Halomonas meridiana and other Bacillus sp. (Prakash et al., 2009).
Large scale production of exogenous enzymes combines the disciplines of microbiology, genetics, biochemistry and engineering with the basic principle, fermentation (Sadhu and Maiti, 2013). Fermentation methods are divided into two categories as Solid State Fermentation (SSF) and Submerged Fermentation (SmF) (Murad and Azzaz, 2010).
The SSF is the cultivation of microorganisms on moist solid substrates, like bran, bagasse, paddy straw and other agricultural waste and paper pulp and SmF utilizes free flowing liquid substrates, such as molasses and broth (Subramaniyam and Vimala, 2012). The SSF is best suited for fermentation techniques involving fungi and microorganisms that require less moisture content while SmF is commonly practiced with microorganisms such as bacteria that require high moisture content. Approximately 90% of the commercial enzymes are produced by SmF method as the method allows better control of the conditions during fermentation. The SSF method develops a tight contact with the insoluble substrate therefore achieving higher substrate concentration for fermentation. Since SSF involves relatively little liquid when compared with SmF, downstream processing from SSF is theoretically simpler and less expensive.
APPLICATION OF ENZYMES
There are several enzyme application methods widely used but the most effective method is yet to be recognized. The application methods vary from a pretreatment of the feed for a period of time before feeding (e.g., silage making, forage harvesting) to application at the time of feeding (application to the hay, in Totally Mixed Rations (TMR), concentrate), even the direct application to the rumen. As enzyme activity strictly depends on the type of feed the enzyme-feed specificity should be given a special consideration when selecting an appropriate method (Hvelplund et al., 2009).
Sutton et al. (2003) reported that favorable responses when enzyme added to the TMR mixture possibly due to the improved intake of digestible organic matter where in same study they found no significant effect when enzyme applied to the concentrate or directly infused to the rumen. Significant improvements in both Dry Matter Intake (DMI) and Dry Matter Digestibility (DMD) were observed in sheep fed with fibrolytic enzyme supplemented guinea grass (Panicum maximum) hay (Beauchemin et al., 2001). As suggested by these studies addition of enzymes prior to feeding enhances ruminal fibre digestion by altering the structure of the feed thereby making it more susceptible to degradation. Yang et al. (2000) reported that fibrolytic enzyme addition to concentrates one month before feeding increased diet digestion and milk production by dairy cows. Enzyme applied at forage harvesting improved the Crude Protein (CP) intake, CP digestibility, DMI and DMD of Bermuda grass hay (Krueger et al., 2008). Hvelplund et al. (2009) found that use of aerobic enzymes in combination with ensiling showed negative effects as the Neutral Detergent Fiber (NDF) content increased and the in vitro digestibility decreased markedly (between 14 and 19% units) compared to the silage without enzyme treatment and irrespective of forage. Another study by Giraldo et al. (2008) demonstrated that supplementing a fibrolytic enzyme directly into the rumen increased the fibrolytic activity in ruminal fluid without a pre-feeding feed-enzyme interaction. Although enzyme supplementation did not affect significantly the concentration of total Volatile Fatty Acid (VFA) in ruminal fluid, molar proportions of propionate increased and acetate: propionate ratio decreased, which might indicate a change in ruminal bacterial populations. But opposite results were obtained by Lewis et al. (1999) and Hristov et al. (2008) with the same supplementation method. Previous research evidences showed that fibrolytic enzyme supplements are more effective when applied to dry versus moist feeds (Feng et al., 1996). The proposed theory behind the positive effects with pre-treatment of enzyme is the enhancement of the binding of enzyme with the feed substrate, which may increase the resistance of the enzymes to proteolysis and prolong their residence time within the rumen.
The method of enzyme application appears to be an important concern in order to obtain positive responses from animal, however further research on this area is strongly suggested.
PRODUCTION RESPONSES IN RUMINANTS
Supplementation of ruminant rations with exogenous enzymes drew a considerable research attention with the successful story of monogastric industries and it is steadily growing up to date. Following content will discuss newer research findings on this field, separately on ruminant species. The noteworthy facts about these researches are that numerous enzyme products have applied at various dose rates to different forage types to different animals in various stages of production and in most of the times results show a higher variability. Despite the immense number of studies conducted, generating a precise conclusion or protocol on enzyme utilization has become a challenge due to several reasons, specially the lack of information on enzyme activity and concentrations.
Dairy cattle: From a study done by Mohamed et al. (2013) they found that supplementation of early lactating dairy cow diet with fibrolytic enzymes (Enzyme was added to the TMR at the time of feeding), did not cause any significant changes in dry matter intake. But with the supplementation of exogenous fibrolytic enzymes milk yield was improved significantly (p<0.003) (41.0 vs. 39.5 kg/cow/day) compared to untreated dairy cows. In addition, the energy corrected milk (40.6 vs. 39.4 kg) and feed efficiency in early lactating dairy cows were improved significantly compared to the control group. These results consistent with outcome of Lopuszanska-Rusek and Bilik (2011) where they observed enhanced milk production with xylanase-esterase supplementation and a tendency of improving DMI and milk production with xylanase and cellulase enzyme supplementation, respectively (Table 2). In contrast to these results Dean et al. (2007) revealed that enzyme supplementation did not improve in vivo digestibility of lactating cows therefore, milk production was not significantly affected (Table 2). Elwakeel et al. (2007) observed that dry matter intake, milk production, milk efficiency, production of milk and milk compositions were not affected by the addition of fibrolytic enzymes (Table 2). But they hypothesized that slight differences of milk production might be due to repartitioning of energy between milk and body reserves for cows receiving enzymes. Similar results, non-significant changes of milk production with the supplementation of direct fed microbials and enzyme mixture was observed by Diler et al. (2014) (Table 2). Significant increment of milk production in response to the enzyme supplementation was observed by Klingerman et al. (2009) where the percentages of milk fat and milk protein were unaffected by treatment (Table 2). Holtshausen et al. (2011) observed no effect on milk yield but dry matter intake was lower for the high enzyme treatment (Table 2). Accordingly, milk production efficiency (kg of 3.5% fat-corrected milk/kg of DMI) linearly increased with increasing enzyme addition.
Apart from fibrolytic enzymes there is evidence that exogenous proteolytic enzyme could increase the total tract digestibilities of DM, Organic Matter (OM), Acid Detergent Fiber (ADF) and NDF with larger increases in digestibility of cows though the feeding of proteolytic enzyme unexpectedly decreased feed intake of cows (Eun and Beauchemin, 2005).
| Table 2: | Livestock species wise response for the exogenous enzyme supplementation (summarized details from studies reviewed) |
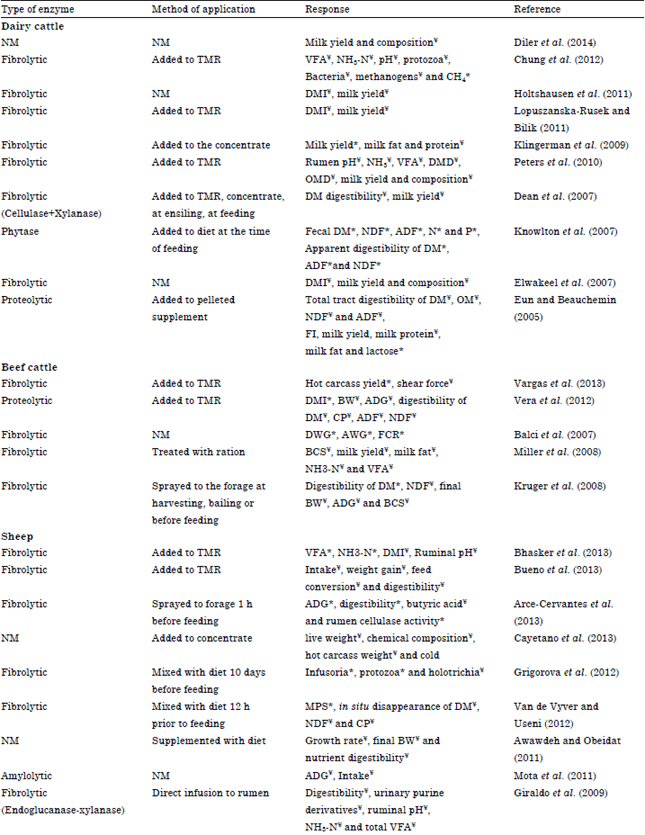  | |
As a result, milk production was suppressed, nevertheless, dairy efficiency, expressed as milk/DM intake, was increased. Supplemented proteolytic enzyme enhanced some milk composition factors such as milk fat and milk lactose percentages but decreased milk protein percentage (Table 2).
As some studies suggested exogenous enzymes, especially phytase can be used to reduce manure nutrient excretion which is environmental friendly. Knowlton et al. (2007) stated that feeding cows with enzyme formulation reduced fecal DM, NDF and ADF excretion and reduced fecal excretion of nitrogen and phosphorous (Table 2).
A study by Chung et al. (2012) disclosed the effect of enzyme additive on VFA production, ammonia nitrogen (NH3-N) flow, pH, or population densities of total protozoa, bacteria and methanogens in ruminal fluid remained unchanged (Table 2). Increasing the level of enzyme supplement in the diet also linearly increased enteric methane (CH4) production, even when adjusted for feed intake or milk production. In a study conducted by Gado et al. (2009), they found that supplementation of dairy cow diet, a TMR with mixture of exogenous enzymes (ZADO®) increased (p<0.05) rumen microbial N synthesis, Short Chain Fatty Acid (SCFA) concentrations, DMI and OMI. Digestibility of all nutrients was higher (p<0.05) in the total tract of supplemented cows. But Peters et al. (2010), examined the influence of fibrolytic enzyme supplementation with TMR prior to feeding on ruminal fermentation, microbial protein synthesis, nutrient digestion and milk yield and composition and there were no significant differences in any of the parameters examined (Table 2).
A meta-analysis (Ortiz-Rodea et al., 2013) was performed to investigate the effect of the addition of exogenous enzymes in ruminant feeding on milk production and chemical composition. After analyzing the observations of 29 experiments which included 52 treatments, 9 enzymes and 1187 animals it was found that the addition of enzyme has no effect on the increment in milk yield production (p = 0.16), fat content (p = 0.88), lactose (p = 0.39) or protein (p = 0.95).
Beef cattle: Vargas et al. (2013) conducted a research to determine the impact of different levels of a fibrolytic enzyme in a finishing diet on steer performance and carcass characteristics. The dietary enzyme levels were 0, 2, 4 and 6 ppm. With the enzyme treatment hot carcass yield improved (linear and quadratic, p<0.05) and the shear force tended to be reduced (linear, p<0.10). No significant differences were detected in gain, feed conversion and loin characteristics concluding that fibrolytic enzymes do not affect steer performance but improve carcass yield and tenderness. In a study by Salem et al. (2011) reported enzyme addition did not affect DM intake, whereas it increased total tract apparent digestibility of nutrients including NDF and ADF, concentrations of rumen ammonia N and total Short Chain Fatty Acids (SCFA) and live weight gain (Table 2).
A study conducted by Balci et al. (2007) exhibited better daily weight gains, total weight gains and feed conversion rates (Table 2). Therefore, there was a possible improvement in production traits with the supplementation of fibrolytic enzyme used in commercial steer’s feed. Gomez-Vazquez et al. (2011) conducted an experiment to evaluate the influence of exogenous fibrolytic enzymes on growth performance and digestion in steers grazing star grass and supplemented with concentrate and sugarcane fermented. Enzyme doses were 0, 15 or 30 g fibrolytic enzyme/kg concentrate. They found that as enzyme level increased, daily gain, intake, digestion and feed conversion were linearly improved allied with the findings of Balci et al. (2007) (Table 2). Challenging the outcome of these two studies, Krueger et al. (2008) stated that enzyme treatments increased (p<0.05) DM digestibility, NH3 and NDF digestibility but not the initial and final Body Weight (BW), Average Daily Gain (ADG), Body Condition Scoring (BCS) and hip height (Table 2).
By a study where steers were supplemented with a proteolytic enzyme they observed the increased DMI (p = 0.02) but had no effects on final BW, BW change and ADG (Vera et al., 2012) (Table 2). Adding proteolytic enzyme during the growing phase decreased NDF digestibility, whereas the digestibility of DM, CP and ADF was not affected. A tendency could be observed for both ADG (p = 0.09) and final BW (p = 0.11) to increase during the finishing phase without affecting BW change which opposed to the growing phase where enzyme increased digestibility (p<0.04) of DM, CP, NDF and ADF. Miller et al. (2008) showed that the enzyme treatment had no effect on total tract OM or fibre digestibility but increased (p<0.05) urinary N excretion (Table 2).
Sheep: Bhasker et al. (2013) conducted a research with supplementation of diets with fibrolytic enzymes (Optimum doses under in vitro conditions were selected). Enzyme combination of cellulase-xylanase 12,800 to 12,800 IU gAlmaraz1 was selected and its effect on feed intake and rumen fermentation pattern were evaluated. The total volatile fatty acids (p<0.01) and ammonia-N concentration was higher in enzyme supplemented group, while no effect was observed on dry matter intake, ruminal pH and total nitrogen concentration (Table 2). Gomaa et al. (2012) utilized exogenous anaerobic bacterial enzyme in a different way by growing green barley on enzyme treated rice straw and then by feeding to Ossimi sheep to investigate digestibility parameters. Adding enzyme rice straw grown barley significantly increased (p<0.05) TDN%, digestibility coefficients, ruminal ammonia-N, total volatile fatty plasma total protein values.
High doses of Exogenous Fibrolytic Enzymes (EFE) were evaluated for their effects on lamb performance (Bueno et al., 2013) and doses were 0, 5 or 10 g of fibrolytic enzyme per 1 kg DM oat straw. Resulting from enzyme treatment intake decreased linearly (p<0.04) with increasing enzyme doses without changing the weight gain, feed conversion, digestibility and ruminal fermentation variables (Table 2). Similar results of unchanged weight gain and dry matter intake were also evident in the study by Torres et al. (2013).
Another study was conducted (Arce-Cervantes et al., 2013) by supplementing a Lignocellulolytic Extract (LE), extracted from the thermo-tolerant basidiomycete to observe the effects on the intake, digestibility, feed efficiency, growth and productive performance of lambs. Average daily gain and digestibility were improved (p<0.05) with LE supplementation (60 and/or 120 mL). Improved cellulase activity in the rumen and a reduction of butyric acid were observed (Table 2).
Meat quality of growing lambs was evaluated with the addition of Salix babylonica L. extracts and exogenous enzymes in combination or individually. They found that only the combination of Salix babylonica extract and enzyme had a significant effect on meat quality parameters but no significant effects with only the enzyme addition (Cayetano et al., 2013). A study by Rivero et al. (2012) reported the effect of Salix babylonica extracts and exogenous enzymes separately and as a combination on haematological parameters in growing lambs where no effect with any treatment observed.
Giraldo et al. (2009) conducted a research work by direct infusion of fibrolytic enzymes with endoglucanase and xylanase activity to the rumen of Merino sheep and found the supplementation with enzyme did not affect diet digestibility (p = 0.30 to 0.66), urinary excretion of purine derivatives (p = 0.34), ruminal pH (p = 0.46), or concentrations of NH3-N (p = 0.69) and total VFA (p = 0.97) (Table 2). Similarly Awawdeh and Obeidat (2011), tested exogenous enzymes on growth performance, nutrient digestibilities and nitrogen balance of male finishing lambs fed diets containing olive cake. Supplemental enzymes had no effects (p = 0.55) on nutrient intakes or growth performance of lambs in terms of growth rate, final BW, total gain, feed conversion ratio and nutrient (DM, OM, CP, NDF and ADF) digestibilities or retained nitrogen. In contrast, molar proportion of propionate were greater (p = 0.001) and acetate: propionate ratio was lower (p<0.001) in enzyme supplemented sheep. In addition, enzyme supplementation tended to increase (p = 0.06) numbers of cellulolytic bacteria at 4 h after feeding (Table 2).
Titi and Lubbadeh (2004) conducted another research with the main objective of investigating the influence of fibrolytic enzyme treatment on birth weight of lambs and they found that birth weight was not significantly affected but the weaning weights of lambs were increased (p<0.05).
The treatment resulted in increased (p<0.05) milk production, total solids, milk, fat and protein percentage with no effect on feed intake (Table 2). Another study conducted by Flores et al. (2008) reported no effects on lactation when the fibrolytic enzyme product was added to the concentrate of dairy ewes.
Increased number of infusoria, protozoa count and reduced rate of Holotricha population in yearling rams fed on diet supplemented with exogenous enzymes were observed (Grigorova et al., 2012) (Table 2).
Supplementation of exogenous fibrolytic enzyme in sheep diet did not change the in situ disappearances of DM, NDF and CP of the roughage mixture but the in situ Microbial Protein Synthesis (MPS) was increased significantly (Van de Vyver and Useni, 2012) (Table 2). Almaraz et al. (2011) observed no marked difference of growth performance of finishing lambs with enzyme treatments (Table 2). Enhanced TDN, CP digestibility with normal blood and rumen parameters in sheep were observed by Gomaa et al. (2012) after treating rations with exogenous enzymes. As suggested by the study of Avellaneda et al. (2009) fibrolytic enzymes reduced duodenal ADF flow and forestomach digestion and there were no enzyme effects on DMI, N balance, ruminal degradation, total tract digestion, ruminal fermentation, as well as ruminal protozoal counts.
Mota et al. (2011) conducted a study to evaluate the effect of exogenous enzyme, glucoamylase from Aspergillus niger addition in finishing diets for lambs. The average daily gain (kg), intake (kg day-1) and were not affected by the treatments (Table 2).
Goat: Shami goats were used in a study by Titi and Lubbadeh (2004) to disclose the influence of fibrolytic enzymes on weaning weight and milk production where they observed significant increment in both parameters with no effect on feed intake. Supplementing dairy goat concentrate with a fibrolytic enzyme mixture enhanced DM and OM in vivo total tract digestibility (Gonzalez et al., 2008) (Table 2). A study by Hussain et al. (2014) witnessed that the enzyme supplementation with the TMR resulted in 31.25% increase in net profit by improving the average daily weight gain significantly (83.49 g in treated group compared with 68.33 g in control) and by a non-significant (p = 0.2875) reduction of feed intake (Table 2). Wahyuni et al. (2012) conducted a research with goats by feeding enzyme supplemented oil palm frond based diet. They observed enzyme supplementation did not affect (p>0.05) Dry Matter Intake (DMI) and average NH3-N concentration but higher ADG and better feed per gain ratio as compared with control. Increased nutrient digestibility was observed in goats fed with enzyme treated diet (Salem et al., 2011) (Table 2). Bala et al. (2009) conducted a research by supplementing enzyme to concentrate portion. There was significant improvement (p<0.05) in the diet digestibility of DM, OM, CP, NDF, ADF and total carbohydrates between the control and treated goats (Table 2). Another study was conducted to investigate the effects of adding cellulolytic enzyme “Asperozym” or Tomoko® to the diets on the performance of goats. The diets supplemented with either enzymes showed significantly (p<0.05) increased digestibility all nutrients for compared with the control diet. Rumen liquor parameters were significantly (p<0.05) affected (Kholif and Aziz, 2014) (Table 2).
Buffalo: Using cellulase, xylanase, alpha-amylase and polygalacturonase enzymes for buffalo calf diets, an experiment was conducted (El-Kady et al., 2006). Feed intake was not affected (p<0.05) by enzyme supplementation but caused a significant (p<0.05) increase in average daily gain, total body weight gain, feed conversion as (kg DM/kg gain) and (kg TDN/kg gain) and Total Digestible Nutrients (TDN) (Table 2).
Malik and Bandla (2010) evaluated the optimum doses of enzymes and probiotics, selected through in vitro experiments on male buffalo calves’ performance. Calves exhibited higher ADG and feed efficiency when fed with mixture of probiotics and enzyme. The OM, NDF and ADF digestibility were improved significantly (Table 2).
An experiment conducted by Thakur et al. (2010) investigate the effect of supplementing exogenous fibrolytic enzymes on the growth performance and nutrient utilization in buffalo calves. High dose of enzyme enhanced the average body weight, average daily gain, DMI and the digestibility coefficient of NDF and cellulose (Table 2).
Supplementation of exogenous enzymes with ruminant diets shows beneficial effects on feed utilization, growth and production performance in ruminant animals though the some debatable issues need to be further revised. Therefore future studies are highly recommended with the special emphasis on feed specific enzyme activity, method of supplementation and optimum dosage of enzymes.
Thulile Sgwane Reply
GOOD PAPER