Research Article
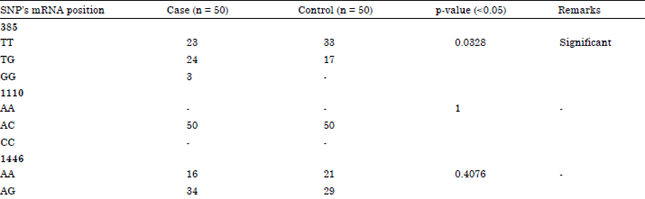
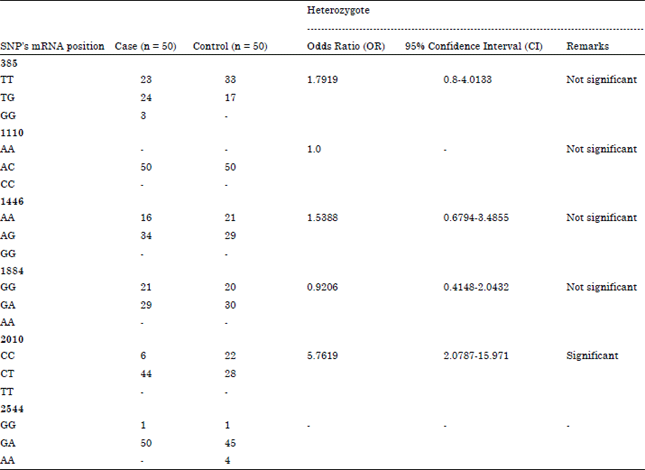
Association between SNPs in TLR2 Gene Segment Corresponding to LRR Functional Domain of TLR2 Receptor and Bovine Mastitis
Department of Veterinary Biochemistry,
J. Thanislass
Department of Veterinary Biochemistry,
Lydia Dhanammal
Department of Animal Genetics and Breeding,
R. Ganesan
Department of Animal Genetics and Breeding,
H.K. Mukhopadhyay
Department of Veterinary Microbiology, Rajiv Gandhi Institute of Veterinary Education and Research, Puducherry, 605 009, India