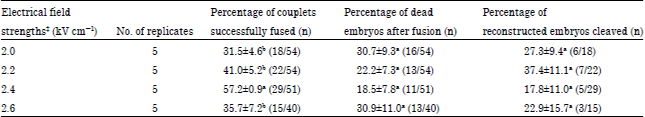
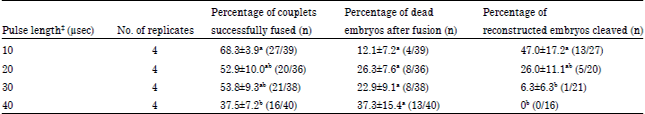
Research Article
Fusion Competence of Interspecies Nuclear Transfer Embryos Reconstructed from Pedicle Periosteum Cell of Sika Deer (Cervus nippon) and Oocyte of Bovine
Institute of Special Animal and Plant Sciences, CAAS, Jilin, 130112, China
Junjun Zheng
Institute of Special Animal and Plant Sciences, CAAS, Jilin, 130112, China
Yuechun Yang
Institute of Special Animal and Plant Sciences, CAAS, Jilin, 130112, China
Zongyue Liu
Institute of Special Animal and Plant Sciences, CAAS, Jilin, 130112, China
Miao Yu
Institute of Special Animal and Plant Sciences, CAAS, Jilin, 130112, China
Chunyi Li
Institute of Special Animal and Plant Sciences, CAAS, Jilin, 130112, China
Fuhe Yang
Institute of Special Animal and Plant Sciences, CAAS, Jilin, 130112, China