Research Article
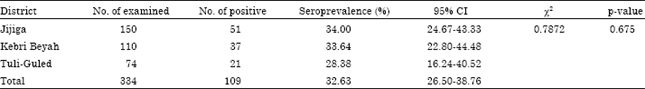
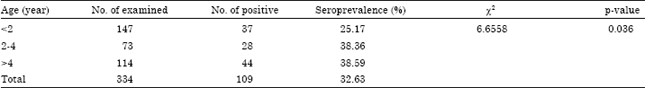
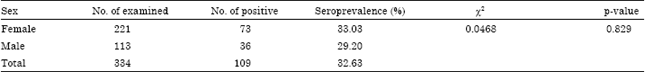
Contagious Caprine Pleuropneumonia: Serological Survey in Selected Districts of Jijiga Zone, Ethiopia
Microbiology and Veterinary Public Health Team, School of Veterinary Medicine, College of Agriculture and Veterinary Medicine, Jimma University, P.O. Box 307, Jimma, Ethiopia
Mekonnen Addis
Microbiology and Veterinary Public Health Team, School of Veterinary Medicine, College of Agriculture and Veterinary Medicine, Jimma University, P.O. Box 307, Jimma, Ethiopia
Mulugeta Tefera
Microbiology and Veterinary Public Health Team, School of Veterinary Medicine, College of Agriculture and Veterinary Medicine, Jimma University, P.O. Box 307, Jimma, Ethiopia










Dr. Mekonnen Addis Reply
Quality contribution to the the novel scientific community which will attract the attention of reputable peers.