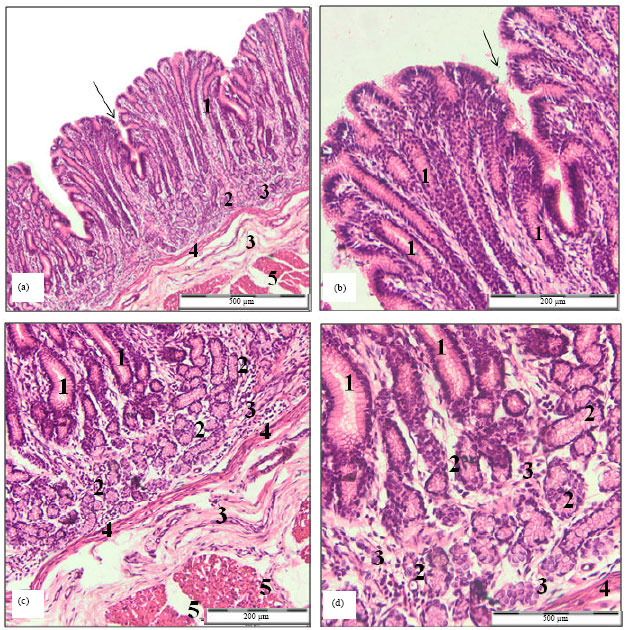
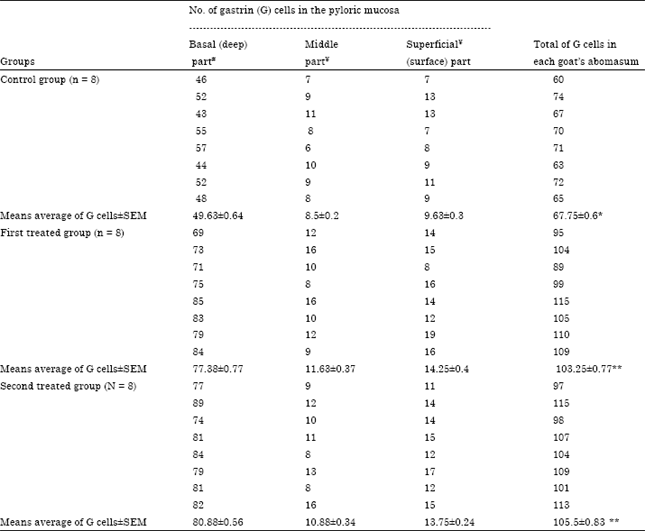
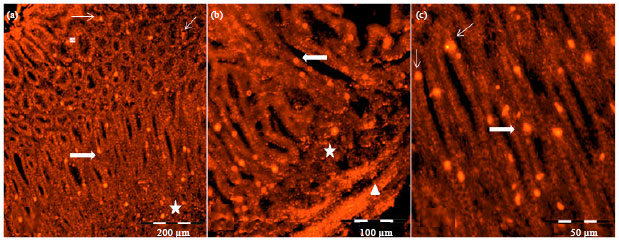
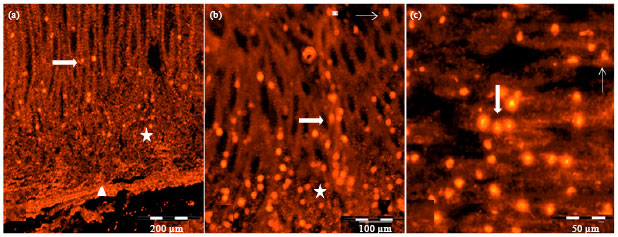
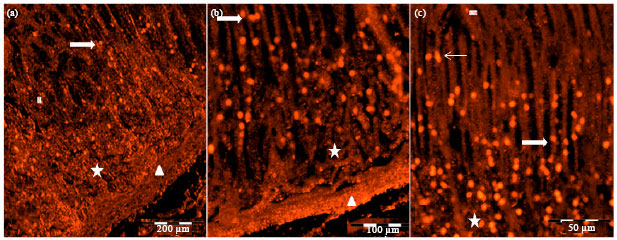
Research Article
Effect of Different Diets on Number and Distribution of Gastrin Cells in the Mucosa of Goat’s Abomasums
Head Section of Histology, Department of Anatomy, Histology and Embryology, Faculty of Veterinary Medicine, Baghdad University, Baghdad, Iraq