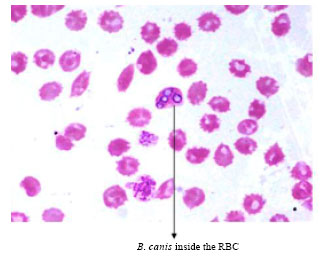
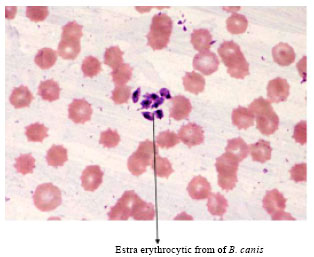
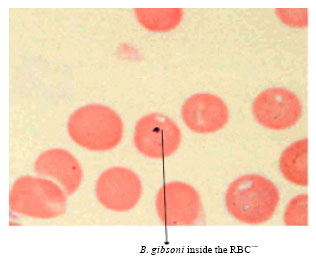
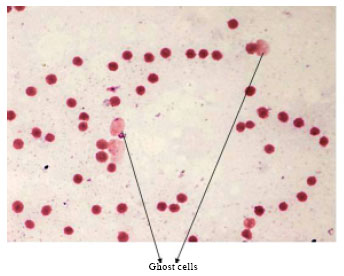
Short Communication
Haemato-biochemical Changes in Natural Cases of Canine Babesiosis
Department of Veterinary Pathology, Guru Angad Dev Veterinary and Animal Sciences University (GADVASU), Ludhiana-141004, Punjab, India
Naresh Kumar Sood
Department of Veterinary Pathology, Guru Angad Dev Veterinary and Animal Sciences University (GADVASU), Ludhiana-141004, Punjab, India
Srinivasa Rao Tumati
Department of Veterinary Public Health, Guru Angad Dev Veterinary and Animal Sciences University (GADVASU), Ludhiana-141004, Punjab, India