Research Article
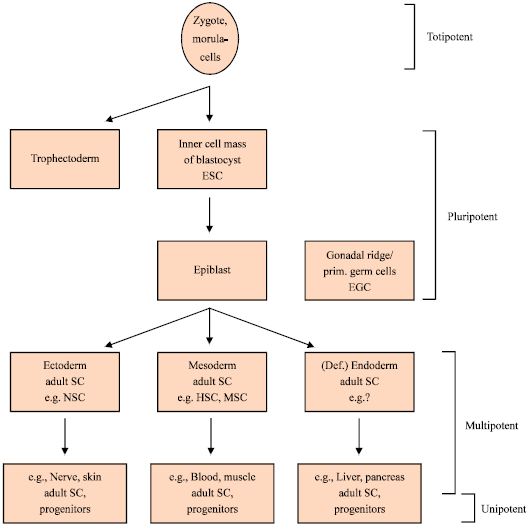
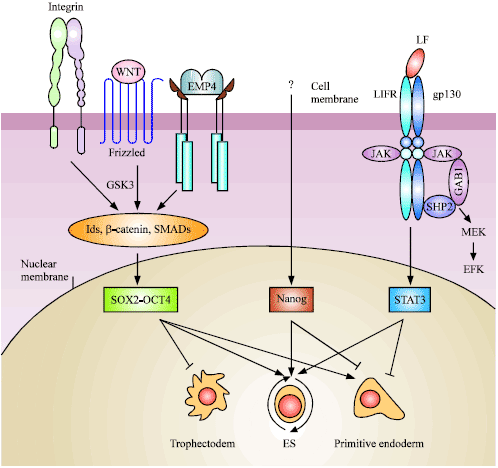
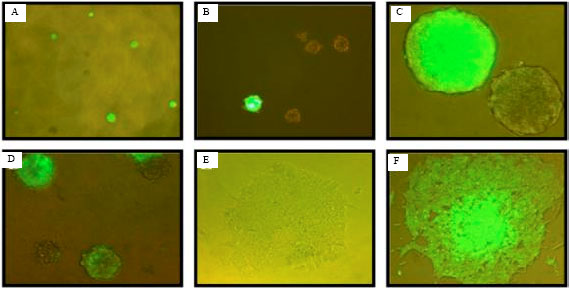
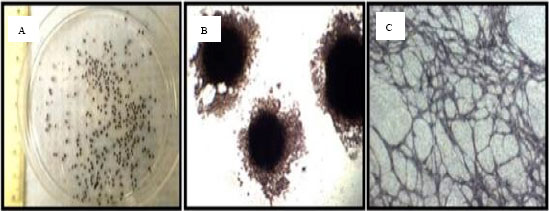
Insights into Embryonic Stem Cells of Bovines
Department of Animal Biotechnology, Centralized Embryo Biotechnology Unit, Tamil Nadu Veterinary and Animal Sciences University, Chennai, Tamil Nadu, India
V. Sejian
Scientist, Division of Physiology and Biochemistry, Central Sheep and Wool Research Institute, Avikanagar, Rajasthan-304501, India
LiveDNA: 91.953
ORCID: 0000-0002-8224-4521