Research Article
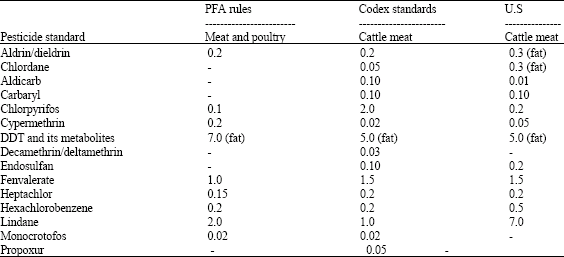
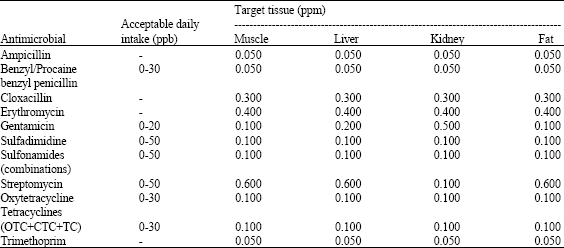
Food Safety Concerns of Pesticides, Veterinary Drug Residues and Mycotoxins in Meat and Meat Products
Department of Livestock Products Technology, Guru Angad Dev Veterinary and Animal Science University, Ludhiana-141004, India
N. Kondaiah
National Research Centre on Meat, Chengicherla, Hyderabad-500 039, India
A.S.R. Anjaneyulu
National Research Centre on Meat, Chengicherla, Hyderabad-500 039, India
P.K. Mandal
Department of Food Science and Biotechnology of Animal Resources, Konkuk University, Seoul, 143-701, S. Korea