Review Article
Mycotoxins in Animal Feeds and Prevention Strategies: A Review
Department of Food Toxicology and Contaminants, Dokki, Giza, Egypt
A.M. Kholif
Department of Dairy Science, National Research Center, Dokki, Giza, Egypt
What Are Mycotoxins?
Mycotoxins are produced by several fungi, particularly by many species of Aspergillus, Fusarium, Penicillium, Claviceps and Alternaria (Bennett and Keller, 1997). Mycotoxins generally display great chemical heterogeneity and approximately 400 of these fungal metabolites are considered to be toxic (Moss, 1996). It is likely that all fungi produce mycotoxins when growth conditions are suitable. Mycotoxins can be carcinogenic, neurotoxic and teratogenic (International Agency for Research on Cancer, 1999; Abdel-Wahhab et al., 1999a, b, 2004). Among the most common mycotoxins are aflatoxins, ochratoxin A (OTA), zearalenone and fumonisins. The first report on human mycotoxicoses dates back to 1100 AD (Gupta and Sharma, 1984). Contamination of diets by mycotoxins and the carryover of mycotoxin related compounds through the food chain (Ramos and Hernandez, 1996) have to be accurately controlled, since minute quantities of mycotoxins are capable of producing allergies, diseases, rashes on skin, neurotoxicity and other disorders.
(a) | |
(b) | |
| Fig. 1: | Exposure of human and animals to mycotoxins. (a) Factors affecting mycotoxin occurrence in the food and feed chain and (b) Cartoon depicting the potential for interactive factors involved in the exposure of animals to mycotoxins |
Although in terms of acute toxicity even the most poisonous of the mycotoxins is far less toxic than the botulinum toxin (Moss, 1996), the consumption of a mycotoxin contaminated diet may induce acute and longterm chronic effects resulting in a teratogenic, carcinogenic (mainly for liver and kidney), oestrogenic, or immunosuppressive impact not only on animals but also on man, whereas animals usually suffer more due to grain of lower quality (Casteel and Rottinghouse, 2000).
Exposure to Mycotoxins
Mycotoxins can enter the food supply in several ways (Fig. 1a, b), but these can be grouped into two general routes of contamination, direct or indirect contamination (Jarvis, 1990). Direct contamination occurs as the result of mold growth on the food material itself. Almost all foods are susceptible to mold growth during some stage of production, processing, storage or transport. Mold growth on foods that are to be consumed directly can result in direct exposure to mycotoxins.
Therefore, the present review was conducted to gain information about contamination of ruminant feeds with mycotoxins, mycotoxins and animal health, strategies for the control of mycotoxins problem and future strategies for mycotoxins management.
LEVEL OF CONTAMINATION OF RUMINANT FEED
Moulds may grow on plants in the field or during the storage period. These fungi may produce toxins, which may have deleterious effects on humans or animals consuming the contaminated product. Such cases of poisoning may cause death in animals, but are rarely fatal in humans (Pfohl-Leszkowicz, 2000). The history of humanity clearly shows that the mycotoxicological risk has existed since the very beginnings of organised agricultural production (Pittet, 1998). Some references to ergotism in the Old Testament (Schoental, 1984) and fusariotoxins such as T-2 toxin and zearalenone are thought to be responsible for the decline of the Etruscan civilization (Schoental, 1991) and the Athenian crisis, which occurred in the Fifth Century B.C. (Schoental, 1994). Certain Egyptian tombs are also thought to contain ochratoxin A, responsible for the mysterious deaths of several archaeologists (Pittet, 1998).
Forages and cereals naturally come into contact with fungal spores before, during and after harvest and during transport and storage. Fungal growth is controlled by a number of physicochemical parameters including the amount of free water (aw), temperature, presence of oxygen, nature of the substrate and pH conditions (Nelson, 1993). Rodents, birds and insects may facilitate contamination by causing physical lesions on plants, providing a route of entry into the plant for fungal spores (Pfohl-Leszkowicz, 2000).
Moulds and Mycotoxins in Fodder
Pastures have been shown to harbour fungi such as Claviceps which are responsible for ergotism, Pythomyces producing sporidesmin which causes facial eczema, Neotyphodium which causes dry gangrene and Rhizoctonia producing slaframin which causes sialorrhea in ruminants (Le Bars and Le Bars, 1996). The mould most frequently encountered in the field belongs to the genus Fusarium. Although this fungus principally infects cereals, it can also be found on forages in the field and after harvest. Depending on the species and environmental factors, Fusarium sp. may produce trichothecenes, zearalenone (ZEN) and/or fumonisins.
Hay harvested in good conditions has a limited and balanced associated microflora, resulting from the successive superimposition of three ecological types of fungi: field fungi (pre-harvest period), intermediate fungi (during harvest) and storage fungi (storage after harvest). Intermediate and storage fungi are more numerous and diverse in hay harvested and stored in humid conditions (Pelhate, 1987). Hydrophilic and heat-tolerant species, such as Aspergillus fumigatus which causes respiratory troubles and could produce gliotoxin and Stachybotrys atra producing satratoxins G and H, which causes stachybotryotoxicosis, both predominate in hay harvested and stored in humid conditions. Numerous highly toxinogenic species of Aspergillus and Penicillium have been detected in damp hay and straw (Clevstroem et al., 1981) as well as Fusarium sp. that produce ZEN (Scudamore and Livesey, 1998). As a consequence, a large range of toxins including the harmful PAT, aflatoxins and sterigmatocystin can be found in insufficiently dried hay and straw.
Moulds and Mycotoxins in Feed Concentrates
Cereals are major mycotoxin vectors because they are consumed by both humans and animals. Between 25 and 40% of cereals world-wide are contaminated with mycotoxins (Pittet, 1998). The most dangerous of these toxins include the aflatoxins produced by Aspergillus flavus and Aspergillus parasiticus. These two fungi are storage moulds often found in cereals, peanuts, cotton and oilseed products from hot and humid countries. They do not affect the plants cultivated in temperate regions, but do affect imported foods. The European Commission set limits of 0.005 ppm for AFB1 in cereals or concentrates. Ochratoxin A (OTA) produced by Penicillium viridicatum may be present in all cereals. It is mainly found in maize, barley, oats, rye and wheat and in oilseed products, particularly if the products were poorly dried before storage. OTA is synthesised after harvest, this phase being the predominant phase of food contamination. Trichothecenes, such as DON, diacetoxyscirpenol (DAS), T-2 toxin and hydroxy-T-2 (HT-2) produced by Fusarium sp., may be present in most cereals during harvest and storage. Fusaric acid which is often present in cereals increases the toxicity of trichothecenes through a synergistic mechanism (Scudamore and Livesey, 1998). Zearalenone (ZEN) is present mainly in maize and also in smaller amounts in sorghum, sesame seeds, barley, wheat and oats harvested late and on grains that have suffered damage to the seed coat. Fumonisins (FB1, FB2, FB3) are principally associated with maize and affect wheat less. The oilseed products, grains and cakes currently used in animal feed may also be contaminated by the three genera of moulds, Aspergillus, Fusarium and Penicillium. However, the mycotoxins produced by these moulds are partly destroyed during oil extraction and are further destroyed during industrial processing.
MYCOTOXINS AND ANIMAL HEALTH
Fungal toxins produce a wide range of injurious effects in animals, in addition to posing food borne hazards to humans (Table 1). The economic impact of decreased productivity, subtle but chronic damage to vital organs and tissues, increased disease incidence because of immune suppression and interference with reproductive capacity is many times greater than that of acute livestock death. Mycotoxins can be classified according to the organ systems they affect. At least one mycotoxin affects each system in the animal body as a direct or indirect mechanism of toxicity. Several important mycotoxins affect the same system, e.g., the immune system and a given mycotoxin may affect several systems concurrently.
Aflatoxins
Aflatoxins are hepatotoxic, hepatocarcinogenic (Fig. 2a, b), mutagenic and teratogenic (Mayura et al., 1998; Abdel-Wahhab et al., 1998, 2002a; Abdel-Wahhab and Aly, 2003, 2005).
| Table 1: | Commodities in which mycotoxins have been found and the resulting effects on animals and humans |
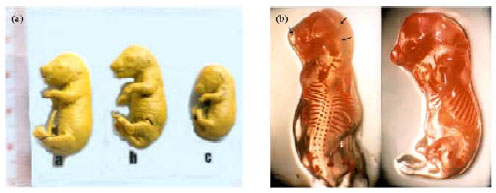
 | |
| Fig. 2: | Effect of aflatoxin on liver and kidney tissues in rats (Abdel-Wahhab et al., 2005c). (a)Liver: congested central vein, damaged bile ducts and massive vacuolar degeneration with abnormal nuclei (shrunken, S., Pyknotic, P.) and (b) Kidney: hyaline casts in proximal and distal tubules lumen and cellular debris Intertubular mononuclear cellular infiltration |
Aflatoxins cause oxidative stress by increasing lipid peroxidation (McKean et al., 2006) and decreasing enzymatic and non-enzymatic antioxidants in aflatoxin-treated rats (Abdel-Wahhab and Aly, 2003, 2005). Acute aflatoxin poisoning provokes major signs of liver lesions, leading to congestion and bleeding (Pier, 1992; Pier and Richard, 1992). Aflatoxicosis causes fatty acid accumulation in the liver, kidneys and heart and may be responsible for encephalopathies and oedemas (Pfohl-Leszkowicz, 2000). The animal may die within a few hours or days. Chronic toxicosis is however more common and, in this case, the liver is also the main target. Aflatoxins act as a DNA intercalating agent by binding to guanine bases and leading to cell death or its trans-formation into a tumour (Riley, 1998).
Ochratoxin A
Although the major target organ is the kidney, OTA has been shown to be hepatonephrotoxic (Kuiper-Goodman et al., 1989), immunosuppression (Fink-Gremmels, 2005), teratogenic (Abdel-Wahhab et al., 1999a), apoptosis (Baldi et al., 2004), genotoxic (Alvarez et al., 2004) and enhancement of lipid peroxidation (Abdel-Wahhab, 2000; Baldi et al., 2004; Abdel-Wahhab et al., 2005a).
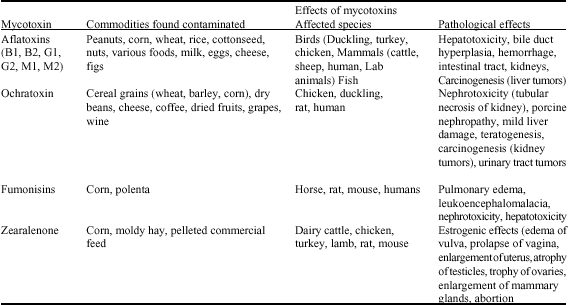
 | |
| Fig. 3: | Fetuses of rats treated with fumonisin (Abdel-Wahhab et al., 2004). (a) severe fetal growth retardation and (b) skeletal malformation |
Ochratoxicoses has been detected in humans in the Balkans and in pigs in Scandinavian countries. This has rarely been found in ruminants because the micro-organisms of the rumen are able to hydrolyse OTA to produce OTAα, which has a lower toxicity. However the detoxification capacity of the rumen may be exceeded in cases of severe poisoning (Ribelin et al., 1978). Acute ochratoxicoses chiefly affects poultry, rats and pigs and is manifested as damage to the kidneys, anorexia and weight loss, vomiting, high rectal temperature, conjunctivitis, dehydration, general weakening and animal death within two weeks after toxin administration (Chu, 1974; Marquardt and Frohlish, 1992). Chronic poisoning induces a decrease in ingestion, polydipsea and kidney lesions. Pigs are particularly sensitive to OTA (Elling and Moller, 1973). Such poisoning has a significant effect for toxin concentrations exceeding 1400 μg kg-1 of feed. OTA has genotoxic properties due to DNA adduct formation (Pfohl-Leszkowicz, 2000). It also has immunotoxic and carcinogenic properties by decreasing the number of natural killer cells responsible for the destruction of tumour cells. Their partial elimination therefore increases the capacity of OTA to induce renal and hepatic carcinomas (Luster et al., 1987). OTA also decreases the activity of phosphoenol pyruvate carboxykinase (PEPCK) and reduces the level of renal neoglucogenesis (Pfohl-Leszkowicz, 2000). In addition, OTA inhibits B and T lymphocytes (Lea et al., 1989).
Fumonisins
Ingestion of fumonisin B1 (FB1), contaminated foods and feeds has been associated with leucoencephalomalacia in horses (Cavret and Lecoeur, 2006), pulmonary oedema in pigs, nephrotoxicity and liver cancer in rats (Gelderblom et al., 2002; Abdel-Wahhab et al., 2002b, 2004). Due to its structural similarity with sphingosine (McKean et al., 2006; Sultan et al., 2006), FB1 interferes with ceramide synthase leading to intracellular accumulation of sphingoid bases which mediate several key biological processes such as cell proliferation and DNA replication. FB1 inhibits biosynthesis of cellular macromolecules (Voss et al., 2006) and it induces lipid peroxidation in both primary rat hepatocytes (Abel and Gelderblom, 1998). FB1 has been shown to induce apoptosis in p53- null mouse embryo fibroblasts (Ciacci-Zanella and Jones, 1999). Abdel-Wahhab et al. (2004) reported that FB1 was proved to be teratogenic and induce fetal growth retardation in rats (Fig. 3a, b).
Zearalenone
Studies in various species (rodents, pigs and monkeys) have shown that ZEN and its metabolites have estrogenic and anabolic activities (Etienne and Dourmad, 1994) and it has cytotoxic effects to mammalian cell cultures (Cetin and Bullerman, 2005). ZEN was associated with hyperestrogenism and several physiological alterations of the reproductive tract in several laboratory animals (mice, rat, guinea-pig, hamster, rabbits) (Creppy, 2002). ZEN ingestion through contaminated feed is associated with decreased reproductive capacity and other hyperestrogenic conditions, such as vaginal swelling, enlargement of mammary glands and testicular atrophy in farm animals (Wannemacher et al., 2000). The strong estrogenic effect of ZEN is due to its competition with 17 β-estradiol in the binding to cytosolic estrogen receptors present in the uterus, mammary gland, hypothalamus and pituitary gland (Abbes et al., 2006). ZEN decreases the rate of survival of embryos in gestating females, decreases the amounts of luteinising hormone (LH) and progesterone production affecting the morphology of uterine tissues, decreases milk production, feminisation of young males due to decreased testosterone production, infertility and perinatal morbidity. Pigs are the most sensitive animals to ZEN poisoning whereas chickens and cattle show lower sensitivities (Coulombe, 1993). ZEN is only produced in very small amounts in natural conditions and probably in insufficient quantities to cause trouble in ruminants (Guerre et al., 2000). ZEN has, however, been shown to cause infertility in grazing sheep in New Zealand (Towers and Sposen, 1988).
The Fate of Mycotoxins in Ruminants
Ruminal Metabolism
Overall, ruminants are more resistant to most mycotoxins than monogastric animals. This suggests that the rumen and its microbial population should play a role in detoxification. The protozoa fraction of the rumen microbial ecosystem seems to be more effective in mycotoxin metabolism than the bacterial fraction, but protozoa are also more sensitive to these toxins (Westlake et al., 1989).
However, bacteria such as Butyrivibrio fibrisolvens, Selenomonas ruminantium and Anaerovibrio lipolytica are able to use T-2 as a source of energy through two enzyme systems (Westlake et al., 1987). Other strains of B. fibrisolvens have been isolated from the rumen and have been shown to be able to degrade derivatives of the toxins DAS, DON, verrucarin A, ZEN and OTA in vitro (Westlake et al., 1989). OTA is metabolised in the rumen to produce phenylalanine and ochratoxin α, which is less toxic than the parent molecule (Chu, 1974). It may also be esterified to produce ochratoxin C of similar toxicity. ZEN is mostly (more than 90%) converted to a-zearalenol, which is about ten times more toxic than the parent toxin and to a lesser extent, to b-zearalenol, which has a low toxicity. Alpha-zearalenol and ZEN can be hydrogenated in the bovine rumen to produce zeranol, an oestrogenic hormone that stimulates animal growth (Kennedy et al., 1998). Aflatoxins are generally poorly degraded in the rumen, with less than 10% degraded for concentrations from 1.0 to 10.0 μg mL-1 (Westlake et al., 1989). Many bacteria are completely inhibited by concentrations of AFB1 below 10 μg mL-1. Thus, this toxin may disturb the growth and metabolic activity of rumen microorganisms. The action of the rumen on toxins may depend on the nature of the diet which may modify the microbial ecosystem and its metabolic activity, but this aspect has not yet been extensively studied.
STRATEGIES FOR THE CONTROL OF MYCOTOXINS PROBLEM
Strategies for the Prevention of Mycotoxins
The prevention of mycotoxins in environment is a big task. In general, prevention of the contamination of fungi and their mycotoxins in agricultural commodities can be divided into these following three levels.
Primary Prevention
The pre-harvest or post-harvest strategy that should be emphasized in a particular year will be dependent on the climatic conditions of that particular year. Unfortunately, avoidance of weather that favours fungal infection is usually outside human control and the combination of high temperature and drought often precludes increased irrigation and consequently adequate mineral nutrition. Conversely, reducing the moisture level in the field at critical periods is equally impossible. Nonetheless, understanding the environmental factors that promote infection, growth and toxin production is the first step in developing an effective plan to minimize mycotoxins in foods and feeds.
There are many new and exciting pre-harvest prevention strategies being explored that involve new biotechnologies. These new approaches involve the design and production of plants that reduce the incidence of fungal infection, restrict the growth of toxigenic fungi, or prevent toxin accumulation. Biocontrols using non-toxigenic biocompetitive agents is also a potentially useful strategy in corn. However, the possibility of recombination with toxigenic strains is a concern. In the case of F. moniliforme in corn, the use of bacterial biocompetitive agents and non-toxigenic F. moniliforme isolates are under development. One interesting approach is the engineering of corn plants to catabolize fumonisins in situ. Typically these approaches require considerable research and development but have the potential of ultimately producing low cost and effective solutions to the mycotoxin problem in corn. Thus, this level of prevention is the most important and effective plan for reducing fungal growth and mycotoxin production. Several practices have been recommended to keep the conditions unfavorable for any fungal growth. These include:
| • | Development of fungal resistant varieties of growing plants |
| • | Control field infection by fungi of planting crops |
| • | Making schedule for suitable pre-harvest, harvest and post-harvest |
| • | Lowering moisture content of plant seeds, after post harvesting and during storage |
| • | Store commodities at low temperature whenever possible |
| • | Using fungicides and preservatives against fungal growth |
| • | Control insect infestation in stored bulk grains with approved insecticides |
Secondary Prevention
If the invasion of some fungi begins in commodities at early phase, this level of prevention will then be required. The existing toxigenic fungi should be eliminated or its growth to be stopped to prevent further deterioration and mycotoxin contamination. Several measures are suggested as follows:
| • | Stop growth of infested fungi by re-drying the products |
| • | Removal of contaminated seeds |
| • | Inactivation or detoxification of mycotoxins contaminated |
| • | Protect stored products from any conditions which favour continuing fungal growth |
Tertiary Prevention
Once the products are heavily infested by toxic fungi, the primary and secondary preventions would not be then feasible. Any action would not be as effective as the practices mentioned above, since it will be quite late to completely stop toxic fungi and reduce their toxin formation. However, some measures should be done to prevent the transfer of fungi and their health hazardous toxins highly contaminated in products into our daily foods and environment. Only a few practices are recommended:
| • | Complete destruction of the contaminated products |
| • | Detoxification or destruction of mycotoxins to the minimal level |
Decontamination Strategies of Mycotoxins
Contaminated mycotoxins in foods and feeds should be removed, inactivated or detoxified by physical, chemical and biological means depending on the conditions.
| Table 2: | Chemical treatment has been used as the most effective means for the removal of mycotoxins from contaminated commodities |
 | |
However, the treatment has its own limitations, since the treated products should be healthsafe from the chemicals used and their essential nutritive value should not be deteriorated. The following methods are suggested to be applied for effective decontamination of some mycotoxins:
Physical Strategies
Mechanical separation, density segregation, colour sorting, removal of the fines or screenings from the bulk shipments of grains and nuts significantly reduce the mycotoxin content of grains. Simple washing procedures, using water or sodium carbonate solution, result in some reduction in concentrations of ZER and FB1 in grains or corn cultures. Gamma irradiation has successfully been used to control ochratoxin levels in feeds (Refai et al., 1996). In some cases, if the mycotoxin levels are known, it is also possible to dilute out the effects of certain contaminated raw materials by blending to produce a final blended feed below the critical level of the specific mycotoxin (Schaafsma, 2002). Such blending of feeds to reduce mycotoxin concentrations is officially permitted, with some limitations, in several countries (Carlson, 2003).
Chemical Strategies
A wide variety of chemicals have been found to be effective (to different extent) against several mycotoxins. The chemicals (Table 2) used fall into the categories of acids, bases (e.g., ammonia, sodium hydroxide), oxidising reagents (e.g., hydrogen peroxide, ozone), reducing agents (e.g., bisulphite, sugars), chlorinating agents (e.g., chlorine), salts and miscellaneous reagents such as formaldehyde. Ammoniation is the method that has received the greatest attention for detoxification of aflatoxin- or ochratoxin-contaminated feeds and has been used success-fully in several countries (Chelkowski et al., 1982). Ammoniation did not show the same efficacy against fumonisins, since no reduction in toxicity was found when ammoniated FB1 was fed to rats in spite of reduction in FB1 content (Chourasia, 2001). Sodium bisulphite has been shown to react with aflatoxins and trichothecenes to form sulphonate derivatives while peroxide and heat enhance the destruction of AFB1 by sodium bisulphite. Formic, propionic and sorbic acids degrade OTA at concentrations ranging from 0.25 to 1% after exposure times of 3-24 h. Destruction of OTA has been observed after treatment with sodium hypochlorite. Treatment of FB1-contaminated corn by heat treatment with Na-HCO3 + H2O2 alone or with Ca(OH)2 gave 100% reduction of FB1 (Palencia et al., 2003). Calcium hydroxide monomethylamine has been used to decontaminate feeds containing T-2 toxin and diacetoxyscirpenol (Trenholm et al., 1989). Ozone treatment has also been used successfully for decontamination of food products (McKenzie et al., 1997). Chemical methods can be used effectively to reduce mycotoxin levels in feeds. However, these chemicals can cause changes in the nutritional proper.
Physicochemical Strategies
Several reports indicated that phyllosilicates clay have the ability to chemisorbs aflatoxin from aqueous solutions (Phillips et al., 1988). Some aluminosilicates bind AFB1 in vitro to varying degrees and form complexes of varying strength with AFB1. The hydrated sodium calcium aluminosilicate (HSCAS) formed a more stable complex with AFB1 than many of the other compounds tested in vitro (Phillips et al., 1988). The HSCAS, bentonite and montmorillonite were found to protect the laboratory animals from the toxic and teratogenic effects of aflatoxins (Abdel-Wahhab et al., 1998, 1999a, 2002a).
Chemoprotection
It is also clear that, in areas where regulations are not enforced, humans are commonly exposed to mycotoxins. In addition, aflatoxin has attracted attention as a chemical weapon and there is military interest in protecting people from exposure either as a precursor to infectious biological weapons or as a panic weapon. Two major avenues of research have been developed to deal with these possibilities. Chemoprotection against aflatoxins has been demonstrated with the use of a number of compounds that either increase an animal`s detoxification processes (Kensler et al., 1993) or prevent the production of the epoxide that leads to chromosomal damage (Hayes et al., 1998). One technical solution is drug therapy, because several compounds, such as oltipraz and chlorophyll, are able to decrease the biologically effective dose (Wang et al., 1999). However, sustained long-term therapy is expensive, may have side effects and is not likely, given the health budgets of developing countries and their other pressing health problems. For the animal feed industries, a major focus has been on developing food additives that provide protection from the toxins. One approach has been the use of esterified glucomanoses and other yeast extracts that provide chemoprotection by increasing the detoxification of aflatoxin (Kensler et al., 1993).
Enterosorption
Another approach has followed the discovery that certain clay minerals can selectively adsorb aflatoxin tightly enough to prevent their absorption from the gastrointestinal tract (Phillips et al., 1988). Whereas many toxins are adsorbed to surface-active compounds, such as activated charcoal, the bonding is not often effective in preventing uptake from the digestive system. Various sorbents have different affinities for aflatoxins and therefore differ in preventing the biological exposure of the animals consuming contaminated foods. There have been several claims for different adsorption agents, but their efficiency in preventing aflatoxicosis varies with the adsorbent (Phillips et al., 1993). With enterosorption, there is also a risk that nonspecific adsorbing agents may prevent the uptake of micronutrients from the food (Mayura et al., 1998). In vitro tests of hydrated sodium calcium aluminosilicates (HSCAS) suggest that there is little other adsorption of micronutrients and Chung et al. (1990) confirmed this result. The use of HSCAS additives in contaminated feeds has proven effective in preventing aflatoxicosis (Fig. 4) in turkeys, chickens, lambs, cattle, pigs, goats, rats and mice (Phillips et al., 2002; Abdel-Wahhab et al., 1998, 2002b, 2005b) and ZER (Abbes et al., 2006). Montmorillonite (EM) also proved to be effective in removing AFB1 and fumonisin from aqueous solution (Aly et al., 2004).
Biological Detoxification
Biological detoxification can be defined as the enzymatic degradation or biotransformation of mycotoxins that can be obtained by either the whole cell or an enzyme system (Bata and Lasztity, 1999).
Aflatoxins
Flavobacterium aurantiacum is able to irreversibly remove AFB1 from both solid and liquid media (Lillehoj et al., 1967). The ability of this microorganism to remove aflatoxins from foods was demonstrated in various food commodities including vegetable oil, peanut, corn, peanut butter and peanut milk. Recent studies indicated that the factor responsible for degradation of AFB1 by the extract of Flavobacterium aurantiacum is an enzyme (or enzymes; Smiley and Draughon, 2000).
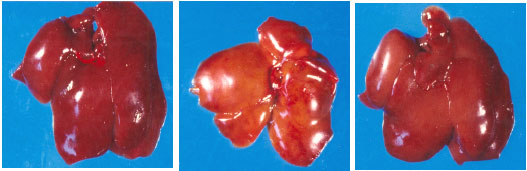 | |
| Fig. 4: | Photograph showing livers of rats treated with aflatoxin alone and in combination with HSCAS or EM. Left: control (normal color of the liver), Middle: aflatoxin treated (pale yellow liver typical of aflatoxicosis), Right: HSCAS or EM plus aflatoxin (normal comparable to the control liver) (Abdel-Wahhab et al., 2005c) |
Other microorganisms including Rhizopus sp., Corynebacterium rubrum, Candida lipolytica, Aspergillus niger, Trichoderma viride, Mucor ambiguous, Neurospora sp., Armillariella tabescens and lactic acid bacteria have been tested in in vitro systems with varying results (Karlovsky, 1999). Most of these microbes convert FB1 to aflatoxicol, which can be converted back to AFB1 by several fungi (by non-aflatoxigenic A. flavus and Rhizopus isolates; Nakazato et al., 1990). Liu et al. (2001) isolated and characterised the enzyme responsible for aflatoxin degradation (the so-called aflatoxin-detoxifizyme) from Armillariella tabescens. This multienzyme complex is possibly responsible for opening the difuran ring of AFB1, thus leading to decreased mutagenic activity.
Ochratoxin A
Of the microorganisms screened for their ability to degrade OTA, several have been found to be able to convert OTA to the far less toxic ochratoxin α. Aspergillus niger could also degrade ochratoxin α to an unknown compound. However, the pathway leading to the opening of the isocoumarin ring is unknown (Varga et al., 2000). Kinetics of OTA detoxification of an atoxigenic A. niger strain and some Rhizopus isolates was also examined. A. niger strain CBS 120.49 could effectively eliminate OTA both from liquid and solid media and the degradation product, ochratoxin α was also decomposed. This atoxigenic A. niger strain, or its enzymes responsible for OTA degradation, could be used for OTA detoxification of agricultural products with low water activities such as cereal grains and green coffee beans. Rhizopus isolates were able to degrade OTA only partially within 10 days. However, only a R. stolonifer isolate could detoxify OTA in spiked moistened wheat. The enzyme involved in the reaction is possibly a carboxypeptidase, as carboxypeptidase A can convert OTA to ochratoxin α (Deberghes et al., 1995; Stander et al., 2001). Further studies are in progress to clone and characterise the carboxypeptidase A gene from Rhizopus isolates.
Fumonisins
Two species of black yeast fungus (Exophiala spinifera, Rhi-nocladiella atrovirens) and a Gram-negative bacterium (Caulobacter sp.) isolated from mouldy corn kernels have been found to extensively metabolise fumonisins to CO2 in liquid culture (Blackwell et al., 1999; Duvick, 2001). These microorganisms produce fumonisin catabolising enzymes, such as esterase which lead to the formation of hydrolysed FB1 (aminopentol 1) plus tricarballylic acid. AP1 has reduced toxicity in comparison to FB1.
Zearalenone
A variety of microorganisms including bacteria, yeasts and fungi are able to convert ZER to α- and β-zearalenol. However, this transformation cannot be regarded as detoxification since the oestrogenic activity of these metabolites is similar to that of zearalenone (α-zearalenol is more oestrogenic, while β-zearalenol is less oestrogenic than zearalenone (Everett et al., 1987). Takahashi-Ando et al. (2002) identified a lactonohydrolase enzyme in the fungus Clonostachys rosea which converts zearalenone to a less oestrogenic compound 1-(3,5-dihydroxyphenyl)-10′-hydroxy-1′-undecen-6′-one.
Antioxidant Strategies
Since some mycotoxins (i.e., AFB1, FB1, OTA and zearalenone) are known to cause cell membrane damage through increased lipid peroxidation (Gautier et al., 2001; Abdel-Wahhab et al., 2004, 2006, 2007), the protective properties of antioxidant substances have been extensively investigated (Galvano et al., 2001). Selenium, some vitamins (A, C and E) and their precursors, have marked antioxidant properties that act as superoxide anion scavengers. For these reasons, these substances have been investigated as protecting agents against toxic effects of mycotoxins (Atroshi et al., 2000). Further evidence of protective effects of some vitamins and/or their precursors against mycotoxin-induced damage arises from numerous in vivo and in vitro studies. Several natural (vitamin, provitamins, carotenoids, chlorophyll and its derivatives, phenolics and selenium) and synthetic (butylated hydroxyanisole and butylated hydroxyl toluene) compounds with antioxidant properties potentially seem to be very efficacious.
Antioxidant and Natural Constituent Defence Against Mycotoxins Toxicity
Feed additives like antioxidants, sulphur-containing amino acids, vitamins and trace elements can be useful as detoxicants (Huwig et al., 2001; Abdel-Wahhab et al., 2004, 2005b, 2006; Abdel-Wahhab and Aly, 2003, 2005). A number of reports are available on the ability of antioxidants to protect from chemical carcinogenesis when they are administered prior to or concomitantly with the carcinogen. The mechanisms behind this protective effect are not completely understood. Addition of numerous antioxidants to foods is often not practical due to economic and labeling restrictions. Furthermore, not all antioxidants are suitable for all foods due to problems associated with color, associated flavors (e.g., herb extracts), solubility and interaction with other food components (e.g., proteins and phenolics). A curious fact is that in some cases the sources of potential protective agents are also a potential way to the assumption of mycotoxins, especially AFB1 and OTA (Halt, 1998). This is the case for coffee, pepper tea, grapes and medicinal herbs. Thus, caution should be used in promoting antimycotoxin action of discussed substances, since some may be carcinogenic (i.e., quercetin) or have toxic (selenium and coumarin) properties. Therefore, preserving or enhancing endogenous oxidation control systems may be more desirable.
FUTURE STRATEGIES FOR MYCOTOXINS MANAGEMENT
Tremendous progress has been made over the last ten years in understanding the factors influencing mycotoxin production and in detecting and diagnosing mycotoxins. However, mycotoxins continue to present a threat to food safety. Changes in agricultural production practices and food processing, along with global changes in environmental and public policy, challenge us to develop and refine strategies and technologies to ensure safe food and a healthy environment. Increasing globalization of trade also adds a new dimension to the importance of mycotoxins not only as toxins, but also as impediments to free trade among countries. An important effect of mycotoxins is the tremendous increase in mycotoxin-related litigation during the past five to seven years. Although some of the law-suits are frivolous, many are not and there has been a marked increase in insurance claims and litigation due to mycotoxins.
From the previous presentation, we addressed the need for an increased ability to diagnose mycotoxicoses in humans and animals. Because of the lack of funding by a number of agencies for animal studies involving mycotoxins, especially long-range, low-toxin-concentration studies in large domesticated animals, there is a void in information necessary to make positive diagnoses for many of the mycotoxins that occur in animal feed. Furthermore, because of the delayed recognition by the medical community of the importance of mycotoxins, there is an even greater lack of information of human intoxications caused by mycotoxins. Presently, considerable information is needed on the association of molds and mycotoxins involved with indoor air quality and human health. Because mycotoxins are unavoidable, they have become one of the leading liability perils in our society involving both the feed and food industry. Therefore, management of the mycotoxin-contaminated matrices is important and adequate testing is the key to any mycotoxin management program. Finally, mycotoxins now must be considered as possible bioterrorism agents. Concern for such use dictates increased understanding about the biosynthesis of these toxins and how they can be rapidly detected and diagnosed.
Listed below are areas of research and public policy that need to be addressed to provide a safer food and feed supply in the twenty-first century.
Critical Needs
With over 500 mycotoxins having been identified the presence of a mycotoxin in a sample is only and indication of contamination as there is a strong likelihood of the presence of other, possibly synergistic, mycotoxins. This is particularly true in mixed feeds where various feed components contribute to the mixture of mycotoxins. This, together with the typical uneven distribution of mycotoxins, requires that maximum acceptable levels be set conservatively in the industry. The control of the problem will however require the commitment from all involved from crop grower through to animal production manager. Generally these critical need cam be summarized in the following points:
Public Policy
| • | Develop uniform standards and regulations for mycotoxin contamination. |
| • | Support joint international cooperation (FAO/ WHO/UNEP) to adopt standardized regulations. |
| • | Develop a safe food supply for local populations. |
Mycotoxin Detection
| • | Develop new technologies for mycotoxin analysis, including multiple-toxin analyses and improve detection (with specificity) of mycotoxins in pre-pared foods. |
| • | Develop biomarkers for human and animal exposure to mycotoxins, including multipanel arrays that can detect exposure to multiple toxins. |
Human and Animal Interactions
| • | Assess mycotoxins as virulence factors. |
| • | Research the effect of mycotoxins as immunosuppressors. |
| • | Evaluate toxicological interactions of toxins with the host (activation and detoxification of mycotoxins by host metabolism). |
| • | Examine population variation for sensitivity to mycotoxins |
| • | Assess interactions among mycotoxins and with drugs, diet and nutrition. |
| • | Assess role of fumonisins on humans and their involvement in esophageal cancer. |
| • | Assess risks of ochratoxin exposure due to its occurrence in a variety of foods and, perhaps, environmental loci. |
Plant and Fungus Interactions
| • | Establish a better understanding of the factors affecting mycotoxin formation in the field and in storage. |
| • | Improve understanding of the ecology and epidemiology of mycotoxin-producing fungi. |
| • | Develop sound agronomic-management practices to decrease mycotoxin contamination. |
| • | Develop host-plant resistance to mycotoxin-producing fungi and to mycotoxin occurrence. |
| • | Develop models to better forecast the potential of mycotoxin contamination. |
| • | Research the genetic regulation and biosynthesis of mycotoxins by the producing organisms. |
Economics of Mycotoxin Contamination
| • | Develop accurate loss estimates for mycotoxin contamination. |
Bioterrorism
Several reports indicated that mycotoxins may have been used in the past as bioterrorism weapons in Southeast Asia. These were described as aerosol attacks in the form of yellow rain with droplets of yellow fluid contaminating clothes and the environment (Wannemacher and Wiener, 1997). With regard to trichothecene mycotoxins, especially T-2, as a biological weapon because they are very stable, resistant to disinfectants, easy to produce in large quantities and can be dispersed through a number of different ways. Additionally, there is strong evidence to suggest that they have been used as biological warfare agents in the past. From this point of view, the following criteria should be considered:
| • | Assess potential for use of mycotoxins as bioterrorism agents. |
| • | Assess mycotoxin-producing fungi as bioterrorism-agent candidates. |
yared addisu Reply
very good article