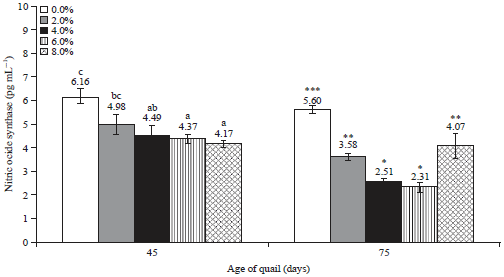
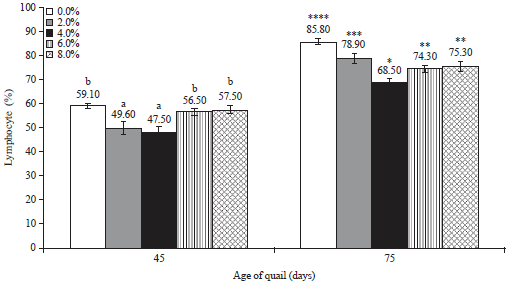
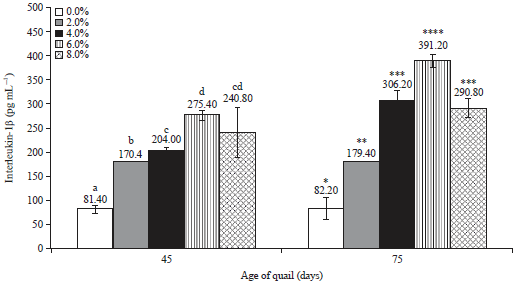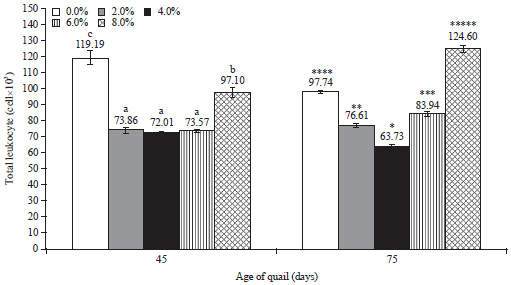Research Article
Egg Cholesterol and Immunity of Quail (Coturnix coturnix japonica) Diet Phillanthus buxifolius Leaves as Feed Supplement
Department of Development Economic, Faculty of Economic, 17 Agustus 1945 University, Surabaya, Indonesia
Jola Rahmahani
Departments of Veterinary Microbiology, Faculty of Veterinary Medicine, Airlangga University, Indonesia
Tatang Sopandi
Departments of Biology, Faculty of Mathematical and Natural Science, PGRI Adi Buana University, Surabaya, Indonesia