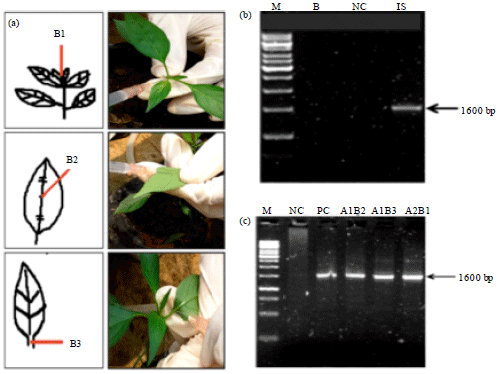
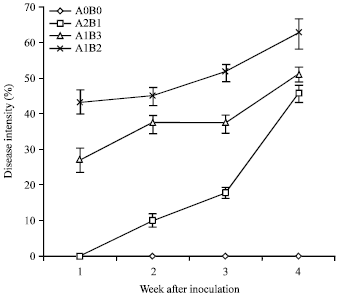
Research Article
Injection Technique Could as a New Promising Method for Artificial Infection of Geminivirus Particles in Chili Pepper (Capsicum annuum L.)
Plant Breeding Section, Faculty of Agriculture, Andalas University, 25163, Padang, West Sumatera, Indonesia
Lily Syukriani
Plant Breeding Section, Faculty of Agriculture, Andalas University, 25163, Padang, West Sumatera, Indonesia
Hashlin Pascananda Utami
Plant Breeding Section, Faculty of Agriculture, Andalas University, 25163, Padang, West Sumatera, Indonesia
Friedrich Herberg
Department of Biochemistry, Kassel University, Heinrich-Plett Str. 40; 34132, Building AVZ I, 2.Stock, Kassel, Germany
Wolfgang Nellen
Department of Genetics, Kassel University, Heinrich-Plett Str. 40; 34132, Kassel, Germany
Istino Ferita
Plant Breeding Section, Faculty of Agriculture, Andalas University, 25163, Padang, West Sumatera, Indonesia