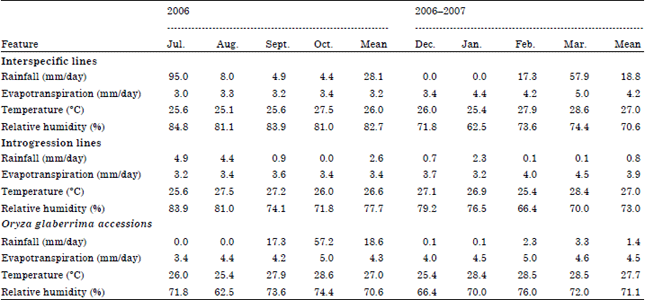
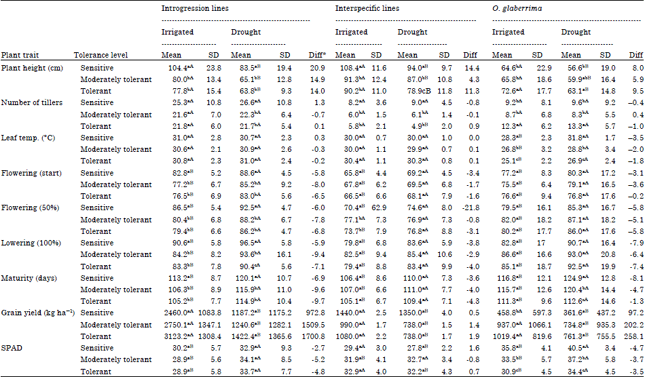
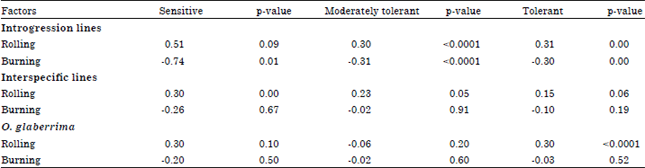
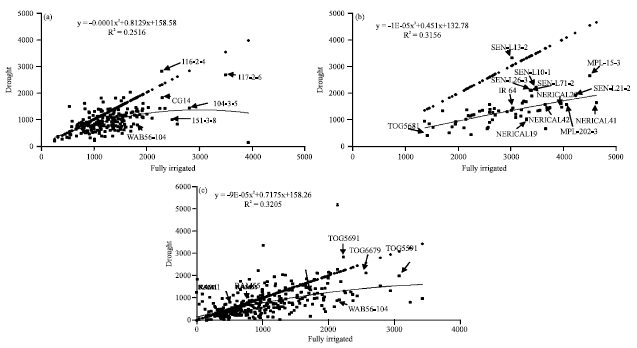
Research Article
Effect of Drought on Oryza glaberrima Rice Accessions and Oryza glaberrima Derived-lines
Africa Rice Center (AfricaRice), 01 B.P. 2031, Cotonou, Benin
P.A. Seck
Africa Rice Center (AfricaRice), 01 B.P. 2031, Cotonou, Benin
M. Lorieux
IRD/CIAT, Agrobiodiversity and Biotechnology Unit, International Center for Tropical Agriculture, AA6713, Cali, Colombia
K. Futakuchi
Africa Rice Center (AfricaRice), 01 B.P. 2031, Cotonou, Benin
K.N. Yao
Africa Rice Center (AfricaRice), 01 B.P. 2031, Cotonou, Benin
G. Djedatin
Africa Rice Center (AfricaRice), 01 B.P. 2031, Cotonou, Benin
M.E. Sow
Africa Rice Center (AfricaRice), 01 B.P. 2031, Cotonou, Benin
R. Bocco
Africa Rice Center (AfricaRice), 01 B.P. 2031, Cotonou, Benin
F. Cisse
Africa Rice Center (AfricaRice), 01 B.P. 2031, Cotonou, Benin
B. Fatondji
Africa Rice Center (AfricaRice), 01 B.P. 2031, Cotonou, Benin