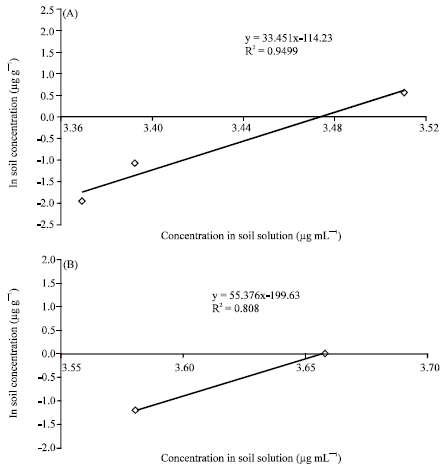
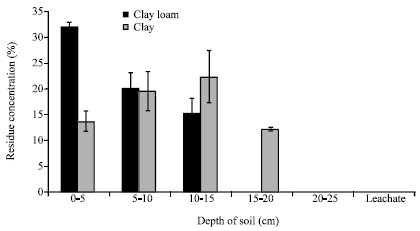
Research Article
Adsorption, Desorption and Mobility of 2,4-D in Two Malaysian Agricultural Soils
School of Environmental and Natural Resource Sciences, Faculty of Science and Technology, Universiti Kebangsaan Malaysia, 43600, UKM, Bangi, Selangor, Malaysia
M. Sameni
School of Environmental and Natural Resource Sciences, Faculty of Science and Technology, Universiti Kebangsaan Malaysia, 43600, UKM, Bangi, Selangor, Malaysia
M. Halimah
Malaysian Palm Oil Board, P.O. Box 10620, 50720, Kuala Lumpur, Malaysia